Calculate DH for the reaction Given the following data: 2NH3(g) + O(g) NH4(1) + HO(1)
Question:
Calculate DH for the reaction
Given the following data:![]()
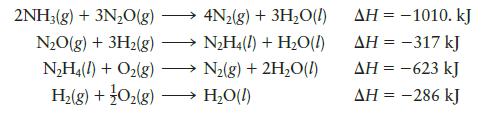
Transcribed Image Text:
2NH3(g) + O₂(g) N₂H4(1) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Answer DH 947 kJ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From the data in Appendix 3, calculate DH for the synthesis of NO (which is the first step in the manufacture of nitric acid) at 25C: 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(l)
-
Calculate DH for the reaction given the following data: NH (1) + O(g) N(g) + 2HO(l)
-
For each data set, calculate s2. (a) (b) (c) -2 3 0 2 9 8 8 9 8 8 9
-
In the figure below, a square of edge lengths is formed by four spheres of masses, m, M, m3, and m4. What is the x component and the y component of the net gravitational force from them on a central...
-
Consider again the conditions of Exercise 7. Describe how to carry out a one-sided Wilcoxon-Mann-Whitney ranks test of the following hypotheses: H0: 0, H1: > 0.
-
In Problem, use derivative formulas to find the derivative of each function. Simplify, except for Problem 10. B = 0.523W - 5176
-
For approximately 20 months, Robert E. McDonald perpetrated a scheme to solicit millions of dollars purportedly for a \($100\) million purchase by the RAI Entities and certain other related corporate...
-
The intangible assets section of Sappelt Company at December 31, 2015, is presented below. Patents ($70,000 cost less $7,000 amortization) ......... $63,000 Franchises ($48,000 cost less $19,200...
-
You have just been named Director of Data Administration of General Hardware Co. General Hardware maintains a large central IS organization with several operational relational databases at its...
-
Apprentice Corporation purchased a pulp and paper mill in Northwestern Ontario on March 1, 2021. The Ontario Ministry of the Environment is requiring that Apprentice Corporation dismantle the mill...
-
Given the following data: calculate DH for the reaction On the basis of enthalpy change, is this a useful reaction for the synthesis of ammonia? (g) + N (g) = 92 kJ (g) = 484 kJ
-
Consider the dissolution of CaCl 2 : CaCl(s) Ca+ (aq) + 2Cl(aq) AH = -81.5 kJ
-
Obtain information pertaining to the U.S. direct investment position abroad on a historical-cost basis from the Bureau of Economic Analysis.:...
-
The business world suffers from a poor public image resulting from unethical behaviors in many organizations. companies are recognizing that good ethical practices make sense and results in business...
-
Belle's Floral Designs is a wholesale shop that sells flowers, plants, and plant supplies. The company operates in a state with no sales tax. The transactions shown below took place during January....
-
Compute the sales forecast for the month of January 2023 using the Simple Moving Average forecasting technique and the last 12 months of data from 2022: Monthly sales for the RUOK Company for the...
-
Plot the magnitude spectrum for the given sequence X(K), then determine the Discrete Fourier transform using direct method. X(K) ={16, 16j,0,-16j, 16,16j, 0, -16j}
-
Task 3: What is the total surplus change caused by a ban on charging different prices?
-
Steam at 80 psia and 400F is mixed with water at 60F and 80 psia steadily in an adiabatic device. Steam enters the device at a rate of 0.05 lbm/s, while the water enters at 1 lbm/s. Determine the...
-
Smiths Family Fashions implemented a balanced scorecard performance measurement system several years ago. Smiths is a locally owned clothing retailer with fashions for men, women, teens, and...
-
For a hydrogen atom in its ground state, calculate the relative probability of finding the electron in the area described. a. In a sphere of volume 1.0 10-3 pm3 centered at the nucleus b. In a...
-
The treatment of a particle in a one-dimensional box can be extended to a rectangular box of dimensions Lx, Ly, and Lz, yielding the following expression for energy: The three quantum numbers nx, ny,...
-
Assume that eight electrons are placed into the allowed energy levels of a cubic box where two electrons can occupy each allowed energy level. (See Exercise 148 for the appropriate energy equation.)...
-
This assignment will make use of a data structure which in Java is called Array List and in C# is called List. Going forward, both will be simply referred to as a list, for simplicity. This...
-
for C++ Write a single statement that perform the following single-subscripted array operations: (a) Initialize the 10 elements of integer array counts to zero. (b) Add 1 to each of the 15 elements...
-
What does the program print? s=0 for n in range(1,5,2): s += n print(s) 10 9 0 4

Study smarter with the SolutionInn App


