Calculate the equilibrium constant at 25C for each of the following reactions, by using data in Appendix
Question:
Calculate the equilibrium constant at 25°C for each of the following reactions, by using data in Appendix 2A: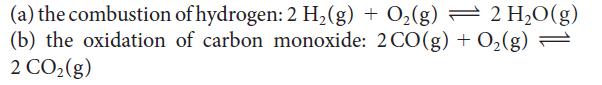
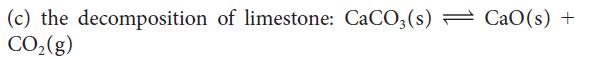
Transcribed Image Text:
(a) the combustion (b) the oxidation 2 CO₂(g) of hydrogen: 2 H₂(g) + O₂(g) — 2 H₂O(g) of carbon monoxide: 2 CO(g) + O₂(g) =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a K 1 10 ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Calculate the standard enthalpy, entropy, and Gibbs free energy at 298 K for each of the following reactions by using data in Appendix 2A. For each case, confirm that the value obtained from the...
-
The budgeted income statement for Barnaby's Hideaway is produced on your Excel spreadsheet. Assume that the following constitute the fixed and vari- able costs for the upcoming year Fixed Costs for...
-
Case1. 640,000 viewers interested in Sports and 360,000 viewers interested in Economy C2 Sports Economy V3 Sports 32,32 36,64 Economy 64, 36 18,18 Questions: 1. What is the equilibrium of the game?...
-
In Exercises 8586, find a. (f g)(x); b. (g f)(x); c. (f g)(3). f(x)=x, g(x) = x + 1
-
A 0.12-F capacitor is given a charge Q0. After 4 s, its charge is Q0. What is the effective resistance across this capacitor?
-
Why do economists focus on historical episodes of hyperinflation to decide whether inflation is a monetary phenomenon?
-
Suppose $X \sim N(0,1)$ and $W_{n} \sim \chi_{n}^{2}$ independently for any positive integer $n$. Let $V_{n}=X / \sqrt{W_{n}} / n$. a. We know $V_{n} \sim t_{n}$. Show that $V_{n}^{2}$ follows an...
-
A group of fraud examiners is coordinating an investigation at a local law firm. Several lawyers at the firm are suspected of overbilling clients, possibly creating fake client accounts, and then...
-
Tax on purchases during the year Interest from a savings account Interest from City of Boston Bonds Dividend from 3 M stock $ 1 , 4 0 0 5 0 0 9 , 0 0 0 2 , 6 0 0 1 , 0 0 0 6 , 2 0 0 8 0 0 7 0 0 4 0 0...
-
The following data are available for Sellco for the fiscal year ended on January 31, 2023: Required: a. Calculate cost of goods sold and ending inventory under the following cost flow assumptions...
-
A reaction mixture that consisted of 0.20 mol N 2 and 0.20 mol H 2 was introduced into a reactor of volume 25.0 L and heated. At equilibrium, 5.0% of the nitrogen gas had reacted. What is the value...
-
Puddings contain large starch molecules that cause the mixture to thicken by a mechanism similar to that by which gelatin thickens. Which of the following suggestions is the best description of how...
-
Match the polar equations with the graphs labeled IIX. Give reasons for your choices. (a) r = cos 3 (b) r = ln , 1 6 (c) r = cos(/2) (d) r = cos(/3) (e) r = sec(/3) (f) r = sec (g) r = 2 , 0 8...
-
A quality-control manager randomly selects 70 bottles of ketchup that were filled on July 17 to assess the calibration of the filling machine. A research objective is presented. For each, identify...
-
Suppose you are conducting a survey regarding violence in hockey in a youth hockey league. You obtain a cluster sample of 10 hockey teams within a youth hockey league and sample all hockey players in...
-
Let \(Y\) be distributed according to the gamma(12 4) distribution. (a) Find \(\mathrm{E}[Y]\). (b) Find \(\operatorname{Var}[Y]\). (c) Find \(P(Y\)
-
Amount of money spent on computers this year Classify the variable as qualitative or quantitative.
-
The average score for a class of 28 students taking a calculus midterm exam was 72%. Determine whether the underlined value is a parameter or a statistic.
-
Modify the spreadsheet in Sidebar 17-4 to compare the purchase of two pieces of equipment based on their rate of return. Test your spreadsheet by entering the data from Problems 8 and 9. Compare your...
-
Research corporate acquisitions using Web resources and then answer the following questions: Why do firms purchase other corporations? Do firms pay too much for the acquired corporation? Why do so...
-
Identify the mechanism expected to operate when 2-bromo-2-methylpentane is treated with each of the following reagents: a) EtOH b) t-BuOK c) NaI d) NaOEt e) NaOH
-
When 1-chlorobutane is treated with ethanol, neither elimination process (E1 or E2) is observed at an appreciable rate; a) Explain why an E2 reaction does not occur. b) Explain why an E1 reaction...
-
Assuming that H o f is constant in the interval 275 K600. K, calculate G o for the process (H 2 O, g, 298 K) (H 2 O, g, 600.K). Calculate the relative change in the Gibbs energy.
-
How can advances in psychoneuroimmunology shed light on the bidirectional communication between the central nervous system and the immune system in the context of stress, and what implications does...
-
You believe that a company will pay a $ 2 dividend per share next year and following this year the dividends will have a dividend growth rate of 6 % annually in perpetuity. You want a 1 2 % yield in...
-
Suppose the real risk - free rate is 3 . 2 5 % and the future rate of inflation is expected to be constant at 2 . 3 0 % . What rate of return would you expect on a 1 - year Treasury security,...

Study smarter with the SolutionInn App


