Carbon dioxide absorbs infrared energy during bending or stretching motions that are accompanied by a change in
Question:
Carbon dioxide absorbs infrared energy during bending or stretching motions that are accompanied by a change in dipole moment (from zero). Which of the transitions pictured of Major Technique 1 on the website of this book can absorb infrared radiation? Explain your reasoning. See Box 8B.1.
Transcribed Image Text:
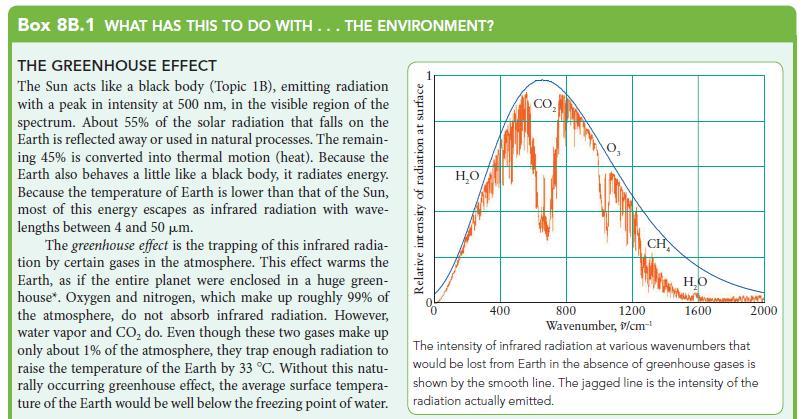
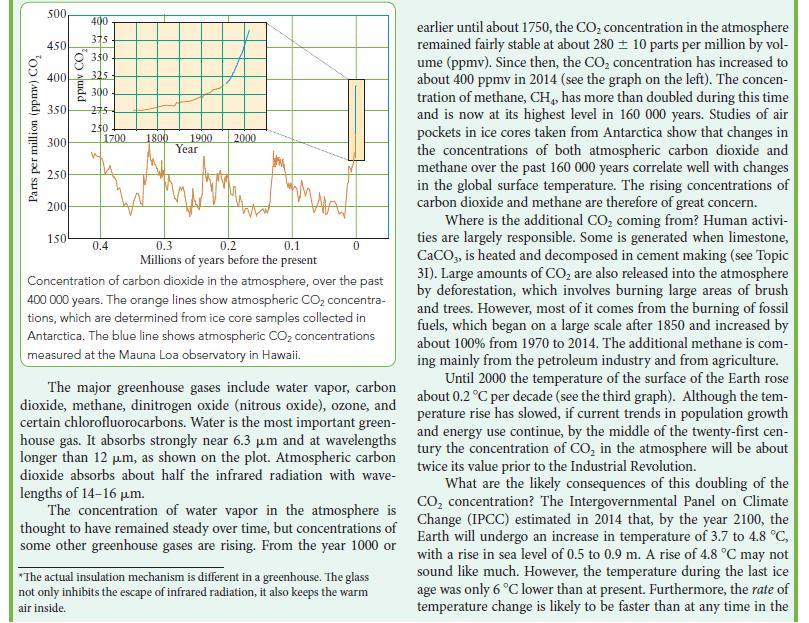
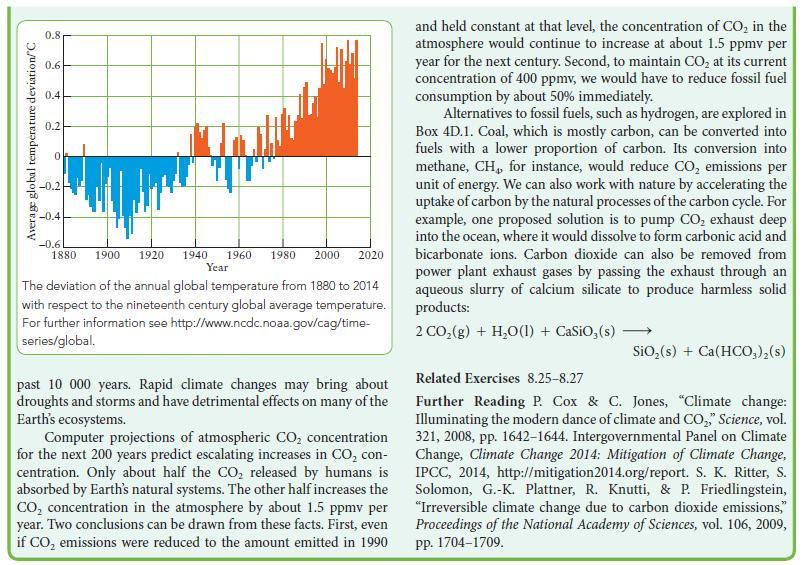
Box 8B.1 WHAT HAS THIS TO DO WITH... THE ENVIRONMENT? THE GREENHOUSE EFFECT The Sun acts like a black body (Topic 1B), emitting radiation with a peak in intensity at 500 nm, in the visible region of the spectrum. About 55% of the solar radiation that falls on the Earth is reflected away or used in natural processes. The remain- ing 45% is converted into thermal motion (heat). Because the Earth also behaves a little like a black body, it radiates energy. Because the temperature of Earth is lower than that of the Sun, most of this energy escapes as infrared radiation with wave- lengths between 4 and 50 m. The greenhouse effect is the trapping of this infrared radia- tion by certain gases in the atmosphere. This effect warms the Earth, as if the entire planet were enclosed in a huge green- house*. Oxygen and nitrogen, which make up roughly 99% of the atmosphere, do not absorb infrared radiation. However, water vapor and CO do. Even though these two gases make up only about 1% of the atmosphere, they trap enough radiation to raise the temperature of the Earth by 33 C. Without this natu- rally occurring greenhouse effect, the average surface tempera- ture of the Earth would be well below the freezing point of water. Relative intensity of radiation at surface HO 400 CO 8 CH 800 1200 Wavenumber, >/cm- HO 1600 MAPATH 2000 The intensity of infrared radiation at various wavenumbers that would be lost from Earth in the absence of greenhouse gases is shown by the smooth line. The jagged line is the intensity of the radiation actually emitted.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Both the antisymmetrical stretch v3 and the doubly ...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
From a taxable income perspective, is it better to be an employee or an independent contractor? Why? How is the determination between what constitutes an employee and an independent contractor made?
-
It is desired to scan the Mediterranean anemia on the gene. For this purpose, a simple DNA You are asked to write a program that will do sequencing. A: 1, G: 2, C: 3, T: 4 random numbers from 1 to 4...
-
Technology World tsad the following revenue and expenses during the month ended July 31, Fees for computer repairs Advertising expense Salaries expense Telephone expenses fees for printer repairs...
-
Create a pseudocode solution for problem 4 using Windows synchronization primitives. Suppose that you had created an OS facility that implemented monitors, but not condition variables. Show hoe to...
-
Maverick Wings, Inc. manufactures airplanes for use in stunt shows. Maverick's factory is highly automated, using the latest in robotic technology. To keep costs low, the company employs as few...
-
How long does it take an ambulance to respond to a request for emergency medical aid? One of the goals of one study was to estimate the response time of ambulances using warning lights (Ho \&...
-
A piece of equipment that cost $32,400 and on which $18,000 of accumulated depreciation had been recorded was disposed of on January 2, the first day of business of the current year. For each of the...
-
K Daniel Hair Stylists' adjusted trial balance follows. Prepare Daniel's income statement for the year ended December 31, 2024. (Click on the icon to view the adjusted trial balance.) Net Income...
-
One method for preparing hydrogen as a fuel from methanol uses a two-step process with the overall reaction CH 3 OH(l) + H 2 O(l) 3 H 2 (g) + CO 2 (g). (a) What is the standard reaction enthalpy of...
-
Methanol, CH 3 OH, is a clean-burning liquid fuel being used as a replacement for gasoline. Calculate the theoretical yield in kilograms of CO 2 produced by the combustion of 1.00 L of methanol (of...
-
Show that it is possible to outrun a light ray, if you're given a sufficient head start, and your feet generate a constant force.
-
Question 1 ACME Widgets produces three products: widgets A, B, and C. The following information is available for each product: Product A B C Selling price per unit $10.00 $15.00 $20.00 Direct...
-
Guideline Choose a tech-based company/industry and provide an overview Identified the changes the company has undergone as a result of the COVID-19 pandemic Discuss supply and demand with respect to...
-
The policy at Nash's Trading Post, LLC is to expense all office supplies at the time of purchase: On the last day of the accounting period, there are $810 of unused office supplies on hand and the...
-
USE THE SHEETS IN THE END FOR YOUR DRAFT CALCULATIONS Section A Question 1: Management Information Systems (MIS) 1) Scenario: Alpha Corporation recently implemented a new MIS to handle large datasets...
-
Finest Frozen Foods Pty Ltd employs Bridget Neville as a salesperson. For Bridget Neville tofulfll her employment activities, she travels all over Brisbane. Finest Frozen Foods purchased acar for...
-
Discuss why Title VII of the 1964 Civil Rights Act was passed.
-
Give the products of the following reaction, where T is tritium: dioldehydrase Ad- CH CH3C-COH CoIII) coenzyme B12
-
Radio station call letters consist of four letters (for example, KUOW). a. How many different station call letters are possible using the 26 letters in the English alphabet? b. Stations west of the...
-
Four bases (A, C, T, and G) appear in DNA. Assume that the appearance of each base in a DNA sequence is random. a. What is the probability of observing the sequence AAGACATGCA? b. What is the...
-
The natural abundance of 13 C is roughly 1%, and the abundance of deuterium ( 2 H or D) is 0.015%. Determine the probability of finding the following in a mole of acetylene: a. H- 13 C- 13 C-H b. D-...
-
Chance Company had two operating divisions, one manufacturing farm equipment and the other office supplies. Both divisions are considered separate components as defined by generally accepted...
-
Consolidated Balance Sheet Working Paper, Bargain Purchase On January 1 , 2 0 2 2 , Paxon Corporation acquired all of the outstanding common stock of Saxon Company for $ 2 billion cash. The balance...
-
Bill Mason is considering two job offers. Job 1 pays a salary of $ 3 8 , 4 0 0 with $ 4 , 9 7 0 of nontaxable employee benefits. Job 2 pays a salary of $ 3 5 , 2 0 0 and $ 6 , 8 2 5 of nontaxable...

Study smarter with the SolutionInn App


