Question: Consider the following compounds and formulas. (The formulas are written in such a way as to give you an idea of the structure.) The boiling
Consider the following compounds and formulas. (The formulas are written in such a way as to give you an idea of the structure.)
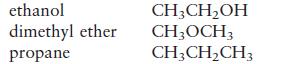
The boiling points of these compounds are (in no particular order) \(-42.1^{\circ} \mathrm{C},-23^{\circ} \mathrm{C}\), and \(78.5^{\circ} \mathrm{C}\). Match the boiling points to the correct compounds.
ethanol dimethyl ether propane CH3CHOH CH3OCH3 CH3CHCH3
Step by Step Solution
3.45 Rating (155 Votes )
There are 3 Steps involved in it
Matching Boiling Points to Compounds Heres the matching of boiling points to the correct compounds B... View full answer

Get step-by-step solutions from verified subject matter experts


