Consider the following illustrations: Which beaker best illustrates what happens when the following acids are dissolved in
Question:
Consider the following illustrations: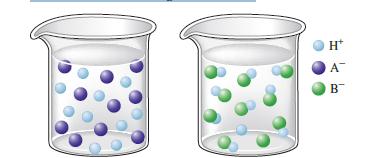
Which beaker best illustrates what happens when the following acids are dissolved in water?
a. HNO2
b. HNO3 e. HC2H3O2
c. HCl
d. HF
e. HC2H3O2
Transcribed Image Text:
H* A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
d HF The beaker on the right with the blue liquid shows what happens when HF ...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following illustrations:
-
Consider the following illustrations: Which beaker best illustrates what happens when the following acids are dissolved in water? a. HNO 2 b. HNO 3 c. HCl d. HF e. HC 2 H 3 O 2 H+ A B
-
The purpose of this problem is to get you used to the concept of autocorrelation in a time series. You could do this with any time series, but here you should use the series of Walmart daily stock...
-
3. Questions A venture capitalist (VC) is willing to invest 100m for 20% ownership of a start-up that is looking to achieve scale. All existing shares are common shares, and this deal would result in...
-
Suppose that the random variable X has the following p.d.f.: Construct a random variable Y = r(X) that has the uniform distribution on the interval [0, 5]. 2e-2 for> o, f(x)-| 2e- for x > 0. 0...
-
A 1.680-g sample of coal contains 1.584 g C. Calculate the mass percentage of C in the coal.
-
In December 2008, Jason Garcia signed a motor vehicle sales contract with Mac Haik Dodge Chrysler Jeep, a dealer. In the contract, Garcia agreed to purchase a 2009 Dodge Ram 1500. The contract...
-
Hall Company had sales in 2014 of $1,560,000 on 60,000 units. Variable costs totaled $720,000, and fixed costs totaled $500,000. A new raw material is available that will decrease the variable costs...
-
1. Application of Legendre transform on van der Waal gas: For monatomic van der Waal gas, the entropy is 3/21 N2a\ S (E,V,N) = Ng In V-Nb 4m E + N 3h2 N 5 V 2 Where KB, m, , h, a, b are constants....
-
For your graduation present from college, your parents are offering you your choice of two alternatives. The first alternative is to give you a money gift of $19,000. The second alternative is to...
-
Is the conjugate base of a weak acid a strong base? Explain. Explain why Cl - does not affect the pH of an aqueous solution
-
You have two solutions of the salts NaX(aq) and NaY(aq) at equal concentrations. What would you need to know to determine which solution has the higher pH? Explain how you would decide (perhaps even...
-
How valuable is the information gathered by RFID technology to a manufacturer/retailer like Serge Blanco?
-
Calculation and discussion of the five-day 1%-Value at Risk (99% confidence level) of your portfolio using model- building approach. Show key steps of workings. (3 marks) Calculation and discussion...
-
Calculate the simple average weighted return per fund on the initial allocation within the portfolio and comment as to why the portfolio return (calculated in #2) is different than the sum of the...
-
Create a GUI that pops up ONLY if the car is selected. This new window should contain JLabels with JTextFields that allow the user to enter in all the car's relevant information - e.g. Make, Model,...
-
Show a GUI calculator program that will offer the user a button to clear the values, buttons for all digits 0 through 9, as well as the operator buttons for multiplication, division, addition,...
-
method ology and Technical Path of "Structual changes in the EU - China Trade --- the impacts of the COVID-19 Pandemic." Create the model which is based on the theory of comparative advantage....
-
Steam expands in an adiabatic turbine from 4 MPa and 500C to 0.1 MPa at a rate of 2 kg/s. If steam leaves the turbine as saturated vapor, the power output of the turbine is (a) 2058 kW (b) 1910 kW...
-
Research corporate acquisitions using Web resources and then answer the following questions: Why do firms purchase other corporations? Do firms pay too much for the acquired corporation? Why do so...
-
The base sequences in mRNA that code for certain amino acids are Glu: GAA, GAG Val: GUU, GUC, GUA, GUG Met: AUG Trp: UGG Phe: UUU,UUC Asp: GAU, GAC These sequences are complementary to the sequences...
-
The change of a single base in the DNA sequence for normal hemoglobin can encode for the abnormal hemoglobin, giving rise to sickle cell anemia. Which base in the codon for glu in DNA is replaced to...
-
The average molar mass of one base pair of nucleotides in DNA is approximately 600 g/mol. The spacing between successive base pairs is about 0.34 nm, and a complete turn in the helical structure of...
-
The population of coyotes in a national forest has consistently increased by 20% each year. This year, the population of coyotes is 5,050. If the population increases at the same rate, what number of...
-
Mark and David are two friends who are both planning to buy a new car. They are choosing between two models of Ford's flagship vehicle: the 2017 Ford Fusion SE and the 2017 Ford Fusion Hybrid. The...
-
After taking this course do you believe your interpersonal communication will improve or remain the same?

Study smarter with the SolutionInn App


