Determine the formal charge on each atom in the following ions. Identify the structure of lowest energy
Question:
Determine the formal charge on each atom in the following ions. Identify the structure of lowest energy in each case.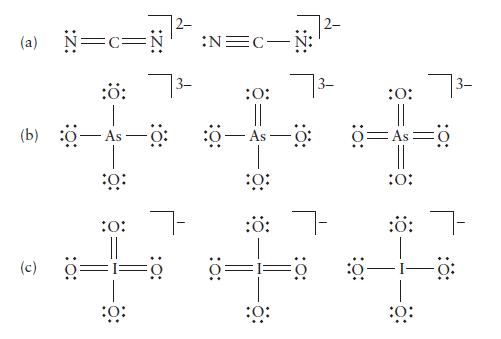
Transcribed Image Text:
(a) N=c= -Ö: (9) (c) :Ö: 0: :0: 7- :Ö: :N=C- :O: As :0: :ö: 7- ö= :0: :O: Ö=As :0: :Ö: :0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine the formal charge on each atom in the given ions well use the formula Formal charge Number of valence electrons in free atom Number of nonbonding electrons Number of bonds Lets apply this ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Determine the formal charge on each atom in the following molecules. Identify the structure of lower energy in each pair. (a) =-: || | :0: H (b) =c=S (c) H-C=N: :0a: T :0: :8-c=s: | H H-CIN
-
Two contributions to the resonance structure are shown below for each species. Determine the formal charge on each atom and then, if possible, identify the Lewis structure of lower energy for each...
-
Create Common Size Analysis (i.e., vertical financial analysis) for Facebook and Twitter This should be a providing an Income Statement Common Size Analysis (CSA) for Facebook and one key competitor...
-
Attlee Ltd holds 28% of the issued shares of Nehru Ltd. Attlee Ltd acquired these shares on 1 July 2019 and on this date all the identifiable assets and liabilities of Nehru Ltd were recorded at...
-
The management of Mid South Utilities Inc. is considering two capital investment projects. The estimated net cash flows from each project are as follows: The generating unit requires an investment of...
-
Briefly define and distinguish between emotional and rational branding.
-
On 1 July 2023, Yananga Ltd, an Australian company that has A$ as its functional currency, enters a loan agreement with a lender in Hong Kong to borrow HK\($700\)000 and uses the funds to acquire...
-
Whitewater Adventures manufactures two models of kayaks, Basic and Deluxe, using a combination of machining and hand finishing. Machine setup costs are driven by the number of setups. Indirect...
-
a) Risk analysis is the study of the underlying uncertainty of a given course of action and refers to the uncertainty of forecasted cash flow streams, the variance of portfolio or stock returns, the...
-
Give the ground-state electron configuration and number of unpaired electrons expected for each of the following ions: (a) Ca 2+ ; (b) In + ; (c) Te 2- ; (d) Ag + .
-
Ionic compounds typically have higher boiling points and lower vapor pressures than covalent compounds. Predict which compound in the following pairs has the lower vapor pressure at room temperature:...
-
What are some arguments for and against each of these policies: dismissal, demotion, return to the same job following rehabilitation, and return to a different job following rehabilitation?
-
Fama and French (2020) proposed two additional models that allow for timevarying risk parameters. Write the equations for these two conditional models. In empirical tests, which model was the best...
-
Fama and French (2015) tested their five-factor model in the sample period 19632013. What test asset portfolios did they use? Was the five-factor model supported?
-
Given their new geometry of the parabola, how did Kolari, Liu, and Huang (KLH) (2021) define the expected returns for the special case of orthogonal portfolios I and ZI on the parabola? Describe...
-
Manufacturing Co. has been negotiating with Imran Elina regarding the sale of some property that represented an old manufacturing site which is now surplus to requirements. Because part of the site...
-
The Greenpeace Report issued in 2020, UNPACKED: How supermarkets can cut plastic packaging in half by 2025, suggested that: GOVERNMENT SHOULD: Set legally binding targets in the Environment Bill to...
-
Suppose that the interest rate is 12% and that the representative agents tastes are such that the interest rate would have to rise to 20% to get him to voluntarily cut current consumption by $1,000....
-
Recall that Chapter 8 described the binary search algorithm for finding a particular entry in an ordered list. The idea behind binary search is to begin looking in the exact center of the list. If...
-
When phosphorus reacts with excess chlorine gas, the compound phosphorus pentachloride (PCl 5 ) is formed. In the gaseous and liquid states this substance consists of PCl 5 molecules, but in the...
-
Describe the molecular structure of the water molecule.
-
Give possible Lewis structures for XeO 3 , an explosive compound of xenon. Determine the formal charges of each atom in the various Lewis structures.
-
A company is considering investing in a new information management system that will cost $2.0 million to buy and $30,000 to install. The system is expected to reduce operating costs by $500,000 per...
-
Below is a two-period price tree of ABC Stock. A European call option on this stock has a strike price of $75 and the risk-free rate is 5% per sub-period. Given that the value of u is 1.22, calculate...
-
Delta Bank plans to issue 10 000, 10-year, 15% coupon bonds. Each bond will be issued at a par value of R1 000. To make the bonds attractive to investors, the bank plans to issue them at a discount...

Study smarter with the SolutionInn App


