Question: Two contributions to the resonance structure are shown below for each species. Determine the formal charge on each atom and then, if possible, identify the
Two contributions to the resonance structure are shown below for each species. Determine the formal charge on each atom and then, if possible, identify the Lewis structure of lower energy for each species.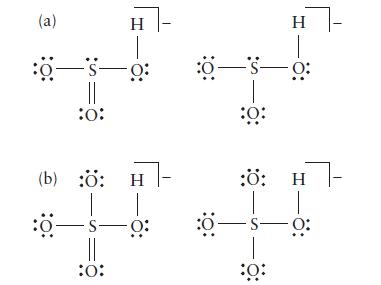
(a) :0-S- 0: :0: H (b) :: H :0-S- :0: H :050: :: :: H 5s0: :: - 0:
Step by Step Solution
★★★★★
3.34 Rating (148 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

a b TO H o OSO 00 l... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


