In which of the following systems is(are) work done by the surroundings on the system? Assume pressure
Question:
In which of the following systems is(are) work done by the surroundings on the system? Assume pressure and temperature are constant.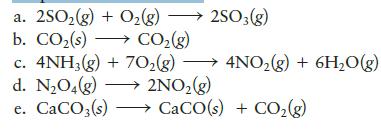
Transcribed Image Text:
a. 2SO₂(g) + O₂(g) →→→ 2SO3(g) b. CO₂ (s) CO₂(g) → c. 4NH3(g) + 70₂(g) d. N₂O4(g) →→→ 2NO₂(g) e. CaCO3(s)→→→→→→ CaCO(s) + CO₂(g) 4NO₂(g) + 6H₂O(g) →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
e CaCO3s CaCOs CO2g In this system work is done by ...View the full answer

Answered By

Sylvanus Oyoo
a business student specifically specialized in the field of accounting
also an IT expert
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In which of the following systems is the energy level separation the largest? (a) A 14Nnucleus in (for protons) a 600 MHz NMR spectrometer, (b) An electron in a radical in a field of 0.300 T
-
Which of the following systems has a unique solution? (a) (b) (c) (d) (e) 022 110 2 xyz 0 3 2 132 130 202 211 132 433 112 0 pqrs 002 250 491
-
Which of the following systems has (i) A unique solution? (ii) Infinitely many solutions? (iii) No solution? In each case, find all solutions: (a) x-2y = 1 3x + 2y = -3 (b) 2x + y + 3z = l x + 4y -...
-
1. As shown by point D in Fig 3.1, the volume of an ideal diatomic gas is 2.00L at standard condition (STP, T=273.15K, P=101.3kPa). The gas is heated to A with its volume conserved, expands...
-
Suppose that a random sample of n observations is drawn from a distribution for which the p.d.f. is as given in Exercise 14. Determine the asymptotic distribution of the sample median.
-
For the following exercises, decompose into partial fractions. x 18 x-12x+36
-
Why is creating a WBS an important aspect of project planning?
-
Tri-County Utilities, Inc., supplies natural gas to customers in a three-county area. The company purchases natural gas from two companies: Southern Gas and Northwest Gas. Demand forecasts for the...
-
Kirpalani Limited wants to expand its furniture line. The board of directors have agreed that the fastest way to raise these funds is to issue bonds. The company proceeds to issue $ 6 0 0 , 0 0 0 , 1...
-
Brike Company, which manufactures one product - robes, has enough idle capacity available to accept a special order of 10,000 robes at $9 a robe. A predicted income statement for the year, without...
-
Which of the following processes are exothermic? a. N(g) 2N(g) b. HO(l) HO(s) c. Cl(g) 2Cl(g) d. 2H(g) + O(g) 2HO(g) e. O(g) 20(g)
-
Nitromethane, CH3NO2, can be used as a fuel. When the liquid is burned, the (unbalanced) reaction is mainly a. The standard enthalpy change of reaction (DH 8 rxn) for the balanced reaction (with...
-
Bhaskar Foods Ltd. acquires a machine whose total cost comes to 270 lacs. The company received a grant of 24 lacs from the central government against the machine. Determine its book value under the...
-
Roseanne is a Scottish taxpayer. She starts trading on 1 July 2020 and prepares accounts for the 16 months to 31 October 2021. The adjusted trading profit for this period is 38,560. Her only other...
-
Joan has been a full-time director of Sunnybank Pursuits Ltd (a trading company) since 2001 and has owned 10% of the company's ordinary shares since 2002. She retired in February 2021 and gave all of...
-
Glenda has been self-employed for many years and prepares accounts to 31 March each year. Her adjusted profits (after deduction of capital allowances) are currently running at approximately 80,000...
-
A company's income statement for the year to 31 March 2021 is as follows: Notes: 1. The company acquired 240,000 of 10% debentures (for non-trade purposes) on 1 January 2021. Interest is receivable...
-
You have been consulted by Mr Christopher Rodrigues on two matters relating to capital gains tax. Extracts from his letter to you are: "On 31 January 2021, I sold my shares in Fledgeby plc, a listed...
-
Discuss some employer tactics used to prevent or minimize union growth prior to the passage of the NLRA in 1935. Which, if any, of these tactics would be lawful today?
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
A galvanic cell is based on the following half reactions: Ag+ + e2 Ag(s) o = 0.80 V Cu2+ + 2e2 Cu(s) o = 0.34 V In this cell the silver compartment contains a silver electrode and excess AgCl(s)...
-
Consider the following galvanic cell: Calculate the Ksp value for Ag2SO4(s). Note that to obtain silver ions in the right compartment (the cathode compartment), excess solid Ag2SO4 was added and some...
-
Consider the following galvanic cell: Ag Cd 1.00 M Ag+ 1.00M Cd2+
-
Jackson, a self-employed taxpayer, uses his automobile 90% for business and during 2023 drove a total of 15,000 business miles. Information regarding his car expenses is listed below. Business...
-
Bel Air Flying Service started operations on December 1 , 2 0 . The trial balance in Figure 1 3 . 6 was prepared at month - end. Record the following adjustments in the General Journal, then make an...
-
Founded in 1960, the Arpeggio Music Company is a family-owned and operated firm specializing in the educational music market. Located in the U.S. Middle West, its products and services include...

Study smarter with the SolutionInn App


