Which of the following processes are exothermic? a. N(g) 2N(g) b. HO(l) HO(s) c. Cl(g)
Question:
Which of the following processes are exothermic?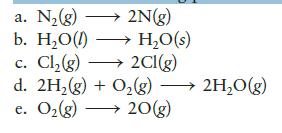
Transcribed Image Text:
a. N₂(g) 2N(g) b. H₂O(l) → H₂O(s) c. Cl₂(g) →→→ 2Cl(g) d. 2H₂(g) + O₂(g) →→→→ 2H₂O(g) e. O₂(g) → 20(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
c Cl2g 2Clg d 2H2g O2g 2HOg Exothermic processes are chemical processes that involve the release of ...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following processes are allowed in electromagnetic interactions and which are allowed in weak interactions via the exchange of a single W or Z 0 ? (a) K + 0 + e + + e , (b) K + + +...
-
Which of the following processes will likely result in a precipitation reaction?
-
Which of the following processes has a AS 0 A) 2H, () O, () 2H,0 (g) B) 2NO2 g) Ba (aq) 2F (aq) 2HgO (s) OB
-
Let F = [F] lb. a) Determine the force Fp acting at roller D. b) Determine the force FE acting at pin E. F 4 ft B 600 lb. ft -3 ft C -3 ft OD -2 ft- E
-
Suppose that a random sample of n observations is drawn from a t distribution with > 2 degrees of freedom. Show that the asymptotic distributions of both the sample mean n and the sample median are...
-
For the following exercises, use any method to solve the nonlinear system. 3x - y = 12 (x - 1) + y = 1
-
Create a simple WBS for planning a wedding.
-
A sphere made of copper having 10 mm inner diameter is placed to an environment at 75C. When the sphere is removed from this environment and exposed to air flow at atmospheric pressure (1 atm). The...
-
The following data which are based on the research conducted by Deakin (1972) present the means of several financial statement items for a sample of 32 failed companies and a matched sample of 32...
-
Jamie Lee Jackson, age 26, is in her last semester of college and is waiting for graduation day that is just around the corner! It is the time of year again when Jamie Lee must file her annual...
-
In a coffee-cup calorimeter, 150.0 mL of 0.50 M HCl is added to 50.0 mL of 1.00 M NaOH to make 200.0 g solution at an initial temperature of 48.28C. If the enthalpy of neutralization for the reaction...
-
In which of the following systems is(are) work done by the surroundings on the system? Assume pressure and temperature are constant. a. 2SO(g) + O(g) 2SO3(g) b. CO (s) CO(g) c. 4NH3(g) + 70(g) d....
-
A rigid, insulated vessel contains superheated vapor steam at 3 MPa, 400C. A valve on the vessel is opened, allowing steam to escape, as shown in Fig. P6.57. The overall process is irreversible, but...
-
On 1 March, Joe Conday started a new business. During March he carried out the following transactions: Required: Draw up a statement of financial position for the business at the end of each day. 1...
-
Consider the example of the three homeowners around the lake (the ones depicted in Table 4.2). Suppose the lake was cleaned up to the efficient level of 2 ppm and that the total costs of the cleanup...
-
Suzanne acquired the following ordinary shares in Quarine plc: She made no further acquisitions and the shares were valued at 3.20 each on 31 March 1982. On 24 July 2020, Suzanne sold 1,200 shares...
-
In May 2015, Ruth sold a freehold building which she had used exclusively for business purposes. The building was sold for 220,000, realising a chargeable gain of 42,500. In the following month, Ruth...
-
Select an agreement from Table 19.1 and investigate the details. For additional readings and websites pertaining to the material inthis chapter, see www.mhhe.com/field8e. Name of Agreement Marine...
-
Discuss some key similarities and differences between AFL and the CIO?
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
Consider the following galvanic cell: Calculate the concentrations of Ag+(aq) and Ni2+(aq) once the cell is dead. Ni 1.0 MNi2+ 10 M Ag
-
A galvanic cell is based on the following half reactions: Fe2+ + 2e Fe(s) o = 20.440 V 2H+ + 2e H2(g) o = 0.000 V In this cell the iron compartment contains an iron electrode and [Fe2+] = 1.00 103...
-
You have a concentration cell with Cu electrodes and [Cu2+] = 1.00 M (right side) and 1.0 1024 M (left side). a. Calculate the potential for this cell at 25oC. b. The Cu21 ion reacts with NH3 to...
-
Explain by giving examples the causes of the differences between taxable income and accounting income ( 5 Marks ) b . Dolphin Company Limited has results of its operations for the year 2 0 1 4 as...
-
Navkar Corporation Limited showed the inventory stock on S ptember 3 0 , 2 0 1 7 , is R$ 7 5 , 0 0 0 . From September 1 5 to September 3 0 , the gross sales of the company were R$ 1 2 5 , 0 0 0 ;...
-
Discuss the process of creating a flexible budget. What are the key steps involved, and how does it differ from the process of developing a static budget?

Study smarter with the SolutionInn App


