Name each of the following organic compounds a. b. c. d. e. CH3CHCHCHCHCHCH
Question:
Name each of the following organic compounds
a.
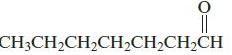
b.
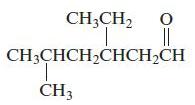
c.
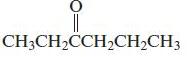
d.
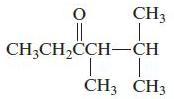
e.
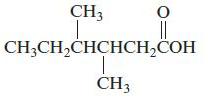
Transcribed Image Text:
CH3CH₂CH₂CH₂CH₂CH₂CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Name of the organic compound are a...View the full answer

Answered By

Danish Ahmad
I have good command on biology as well chemistry from my school days. As I have prepared for NEET and appeared in NEET 2019. I get enrolled in bachelor of pharmacy in year 2019 and now I have good command on biology(cell biology, genetics, cardiology, pharmacology, human physiology, biotechnology, microbiology, biochemistry etc) and chemistry(analytical chemistry, medicinal chemistry, physical, inorganic and organic chemistry) as well as other subjects of pharmacy also.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name each of the following compounds:
-
Name each of the following compounds according to substitutive IUPAC nomenclature: (a) (CH3)2CHCH2CH2CH2Br (b) (CH3)2CHCH2CH2CH2OH (c) Cl3CCH2Br (d) Cl2CHCHB (e) CF3CH2OH
-
Name each of the following using IUPAC nomenclature: (a) (CH3)2CoeC (CH3)2 (d) CH2 = CHCH2 CHCH3 (b) (CH3)3CCHoeCH2 (e) CH2 = CHCH2CHCH3 (c) (CH3)2CoeCHCH2CH2CH3
-
You are deciding between two mutually exclusive investment opportunities. Both require the same initial investment of $10 million. Investment A will generate $2 million per year (starting at the end...
-
A pharmaceutical company wants to test the effectiveness of a new allergy drug. The company identifies 250 females ages 30 to 35 who suffer from severe allergies. The subjects are randomly assigned...
-
Assume that your single aunt Zelda becomes very ill and makes a gift of $ 1 3 , 0 0 0 , 0 0 0 to you. Assuming that she has not made any previous taxable gifts, calculate the gift tax that she will...
-
LVDT transducer a. produces light pulses intermittently b. has high output and is insensitive to temperature c. frequently used in velocity pickups d. has high sensitivity and frequency range e....
-
1. How effective do you think MyGofer will be as a retail venture? 2. In what way is Sears Holding a combination of the old economy and the new economy? 3. What advice can you offer Sears to make...
-
Tinder Confections produces fudge truffles in 10-pound batches. Each batch takes 0.15 hours of direct labor, which includes allowances for breaks, cleanup, and other downtime. Tinder pays its direct...
-
The 2018 balance sheets and income statement for Netflix Inc. follow. Refer to these financial statements to answer the requirements. NETFLIX INC. Consolidated Statement of Earnings For Year Ended...
-
Name each of the following alkenes and alkynes. a. b. c. d. e. f. CH3 CH3CH-C=CH
-
A mist cooler is used to provide relief for a fatigued athlete. Water at T i = 10C is injected as a mist into a fan airstream with ambient temperature of T = 32C. The droplet diameters are 100 m....
-
In this chapter, we introduced normal matrices as those matrices that commute with their Hermitian conjugate. This seems like a weak requirement, but it's very easy to construct matrices for which...
-
When a company assigns goodwill to a reporting unit acquired in a business combination, it must record an impairment loss if a. The fair value of the net identifiable assets held by a reporting unit...
-
Compare the different responsibilities of the accounting and finance departments, and determine if there are any responsibilities that can be eliminated or any that may need to be added.
-
You want it to be something you can do while youre still a student. Research on the Web ideas for small businesses for students. Describe at least three different ideas and include what form of...
-
Discuss the role and functions of The Joint Commission. How do the standards they set aff ect the fi nances and operation of the hospital?
-
Look on the Internet for a current example of a business merger, takeover, or acquisition. Explain the circumstances of the event. What companies are involved? Was the event friendly or unfriendly?...
-
Burlington Company had the following budgeted amounts for production, sales, and costs in April and August, 20A, which are considered to be typical months: Required: (1) Compute the variable overhead...
-
The Adjusted Trial Balance columns of a 10-column work sheet for Webber Co. follow. Complete the work sheet by extending the account balances into the appropriate financial statement columns and by...
-
a. Use the following data to calculate the enthalpy of hydration for calcium chloride and calcium iodide. b. Based on your answers to part a, which ion, Cl2 or I2, is more strongly attracted to...
-
Although Al(OH)3 is insoluble in water, NaOH is very soluble. Explain this difference in terms of lattice energies.
-
Which ion in each of the following pairs would you expect to be more strongly hydrated? Why? a. Na+ or Mg2+ b. Mg2+ or Be2+ c. Fe2+ or Fe3+ d. F- or Br- e. Cl- or ClO4- f. ClO4- or SO4 2-
-
Discuss (1) your own results below. Are you motivated by achievement, power, or affiliation? Is there anything that surprised you? How do you think your motivations influence the way that you lead or...
-
Think of a position you currently have or had. Using the Expectancy Questionnaire, and the Analysis Form,rate each of the three areas on the dimension. For example, for item one think of the amount...
-
1. Compare and contrast the role of the project manager and the functional or operational managers. 2. What is the role of the project manager in terms of responsibility, accountability and authority...

Study smarter with the SolutionInn App


