Name the following compounds. a. b. c. (mathrm{HCOOH}) Cl- || C-OH
Question:
Name the following compounds.
a.
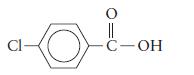
b.
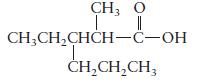
c. \(\mathrm{HCOOH}\)
Transcribed Image Text:
Cl- || C-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Youve provided three compounds to name one as a text formula c and two as structural diagrams a and ...View the full answer

Answered By

HABIBULLAH HABIBULLAH
I have been tutor on chegg for approx 5 months and had solved a lot of questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds by IUPAC rules: a. b. H-C CH,CH-CH
-
Name the following compounds by the IUPAC system: a. CH3CH=C(CH2CH2CH3)2 b. (CH3)2CHCH"CHCH3 c. g. CH3-C-C-CH-CH, h. k.
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
You have a net income of $40 000 per year. Your expenses include the following: Rent: $800 per month Insurance: $225 per semi-annually Car Payment: $315 per month Car Expenses: $1 000 per year ...
-
Evaluate the integral and interpret it as a difference of areas. Illustrate with a sketch. Sm/2 48. 47. 'a sin x dx Ja/4
-
Brilliant Equipment purchased machinery from a Japanese firm and must make a payment of 313.25 million in 45 days. The bank quotes a forward rate of 103.01/$ to buy the required yen. What is the cost...
-
Robert Shapiro was the owner and CEO of Woodbridge, a supposed investment firm. Woodbridges main business model was to solicit money from individuals to invest in low-risk and conservative...
-
Payroll Tax Entries Below is a payroll sheet for Otis Import Company for the month of September 2010. The company is allowed a 1% unemployment compensation rate by the state; the federal unemployment...
-
Assume that Twigs has hired you as a database consultant to develop it's operational database having the three tables described at the end of Chapter 6. Assume that Twigs personnel are the owner, an...
-
Salicylic acid has the following structure: Since salicylic acid has both an alcohol functional group and a carboxylic acid functional group, it can undergo two different esterification reactions...
-
Minoxidil \(\left(\mathrm{C}_{9} \mathrm{H}_{15} \mathrm{~N}_{5} \mathrm{O}ight)\) is a compound produced by the Pharmacia \& Upjohn Company that has been approved as a treatment for some types of...
-
Amber Company produces custom framing. For one job, the dimensions of the picture were such that the computer-controlled, mat-cutting device could not be used. Amber warned the customer that this was...
-
Calculating Annuities You are planning to save for retirement over the next 30 years. To do this, you will invest 500 a month in a share account and 500 a month in a bond account. The return of the...
-
Future Value What is the future value in 4 years of 1,000 invested in an account with a stated annual interest rate of 10 per cent, (a) Compounded annually (b) Compounded semi-annually (c) Compounded...
-
When a new, faster computer chip is introduced, demand for computers using the older, slower chips decreases. Simultaneously, computer makers increase their production of computers containing the old...
-
The following graph shows inflation and unemployment rates for Canada for the period between 1970 and 2012. Does this graph show evidence in favor of the Phillips curve? 14 12 10 8 6 4 1 O 2 Canada...
-
Assuming that Okuns law is given by U U n = 0.75 (Y Y P ) and that the Phillips curve is given by = e - 0.6 (U U n ) + , a) Obtain the short-run aggregate supply curve if expectations are...
-
Green & Freedman Baking Company (Green & Freedman) was a family-owned Massachusetts corporation that produced and sold baked goods. The terms of a collective bargaining agreement required Green &...
-
From the choice of simple moving average, exponential smoothing, and linear regression analysis, which forecasting technique would you consider the most accurate? Why? please write it in word...
-
Describe the structure of the formaldehyde molecule, CH 2 O, in terms of hybrid orbitals, bond angles, and - and -bonds. The C atom is the central atom to which the other three atoms are attached.
-
Noting that the bond angle of an sp 3 hybridized atom is 109.5 and that of an sp 2 hybridized atom is 120, do you expect the bond angle between two hybrid orbitals to increase or decrease as the...
-
Draw the complete Lewis structure for each of the following compounds: (a) Ammonium chloride; (b) Potassium phosphide; (c) Sodium hypochlorite.
-
What are the distinguishing characteristics of transformational delegation, and how does this approach differ from traditional top-down delegation models in driving innovation and organizational...
-
The treasurer of a major U.S. firm has $35 million to invest for three months. The interest rate in the United States is .78 percent per month. The interest rate in Great Britain is 0.6 percent per...
-
A(n) 70-g ice cube at O C is placed in 810 g of water at 28 o C. What is the final temperature of the mixture?

Study smarter with the SolutionInn App


