Oxidation of aldehyde yields a carboxylic acid: Draw the structures for the products of the following oxidation
Question:
Oxidation of aldehyde yields a carboxylic acid:
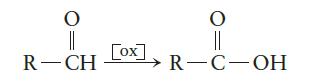
Draw the structures for the products of the following oxidation reactions.
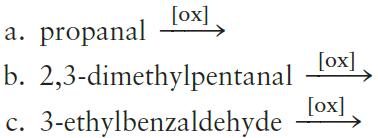
Transcribed Image Text:
O 37 R-CH [ox] O R-C-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Structures of product are as follow 1 Propanal on oxidation ...View the full answer

Answered By

Danish Ahmad
I have good command on biology as well chemistry from my school days. As I have prepared for NEET and appeared in NEET 2019. I get enrolled in bachelor of pharmacy in year 2019 and now I have good command on biology(cell biology, genetics, cardiology, pharmacology, human physiology, biotechnology, microbiology, biochemistry etc) and chemistry(analytical chemistry, medicinal chemistry, physical, inorganic and organic chemistry) as well as other subjects of pharmacy also.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Oxidation of an aldehyde yields a carboxylic acid: Draw the structures for the products of the following oxidation reactions. a. b. c. [ox] propanal 2,3-dimethylpentanal ox] 3-ethylbenzaldehyde>
-
Draw the structures for dichloromethane (also called methylene chloride), CH2Cl2, and trichloromethane (chloroform), CHCl3.
-
Draw the structures for ortho-xylene and meta-xylene.
-
Inferring Investing and Financing Transactions and Preparing a Balance Sheet During its first week of operations ending January 7, 2011, FastTrack Sports Inc. completed six transactions with the...
-
The hourly earnings (in dollars) of a sample of 25 railroad equipment manufacturers Use technology to (a) Find the data sets first, second, and third quartiles, (b) Draw a box-and-whisker plot that...
-
The spacecraft P is in the elliptical orbit shown. At the instant represented, its speed is v = 13,244 ft /sec. Determine the corresponding values of r, , r, and . Use g = 32.23 ft /sec 2 as the...
-
Using an incremental internal rate of return (IRR) analysis, the decision to replace the "current best" by the "challenger"' is based on what decision rule? a. The internal rate of return of the...
-
Rosie Dry Cleaning was started on January 1, 2016. It experienced the following events during its first two years of operation: Events Affecting 2016 1. Provided $45,000 of cleaning services on...
-
A fleet of vehicles is comprised of 60 vans, 20 limos, and X sedans. If 10% of all vehicles are limos, how many sedans are in the fleet?
-
Write an algorithm called SortedSequentialSearch that will return the same results as SequentialSearch but will run more quickly because it can stop with a failure the moment it finds that the target...
-
Polyaramid is a term applied to polyamides containing aromatic groups. These polymers were originally made for use as tire cords but have since found many other uses. a. Kevlar is used in bulletproof...
-
Using appropriate reactants, alcohols can be oxidized into aldehydes, ketones, and/or carboxylic acids. Primary alcohols can be oxidized into aldehydes, which can then be oxidized into carboxylic...
-
Explain how special relativity differs from general relativity.
-
9 Rollins Corporation is estimating its WACC. Its target capital structure is 20 percent debt, 20 percent preferred stock, and 60 percent common equity. Rollins' beta is 1.7 the risk-free rate is 4...
-
What is the purpose of the alt attribute in the img tag? Question 1 0 1 0 options: specify an alternate address for accessing the image specify an alternate image if the image is not found specify an...
-
Rollins Corporation is estimating its WACC. Its target capital structure is 20 percent debt, 20 percent preferred stock, and 60 percent common equity. Its bonds have a 6.8 percent coupon, paid...
-
Two different products are manufactured in a business and they are called as T 1 and T 2 . In a period 5 . 3 5 0 Kg . T 1 and 4 . 6 5 0 Kg . T 2 are produced in a period where the total of the...
-
Lime Industries, Inc. uses 97,820 units of inventory every year. It generally takes seven days for the company to order and receive new inventory. Lime likes to keep an additional seven days of...
-
The Agency for Healthcare Research and Quality reports that medical errors are responsible for injury to 1 out of every 25 hospital patients in the United States. (Data extracted from M....
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
For a simple cubic array, solve for the volume of an interior sphere (cubic hole) in terms of the radius of a sphere in the array.
-
Consider the following melting point data: Account for the trends in melting points for the two series of compounds in terms of interparticle forces. Compound NaCI MgCl AICl SiC PCl SC2 CIh mp (C)...
-
The structures of another class of high-temperature ceramic superconductors are shown below. a. Determine the formula of each of these four superconductors. b. One of the structural features that...
-
Match the event to the expected exchange rate effect between Japan (whose currency is the yen) and South Africa (whose currency is the rand). Assume each has a floating exchange rate policy. 1....
-
Suppose France imports more from Britain than Britain imports from France (because the French really like English bread and cheese!) What type of Balance of Payments imbalance is this?
-
One of the two major airlines serving a city has decreased the price of its fights. What impact wil his have on demand for car rentals in the city? Willingness to pay for car rentals will increase by...

Study smarter with the SolutionInn App


