Question: Polyaramid is a term applied to polyamides containing aromatic groups. These polymers were originally made for use as tire cords but have since found many
Polyaramid is a term applied to polyamides containing aromatic groups. These polymers were originally made for use as tire cords but have since found many other uses.
a. Kevlar is used in bulletproof vests and many high strength composites. The structure of Kevlar is
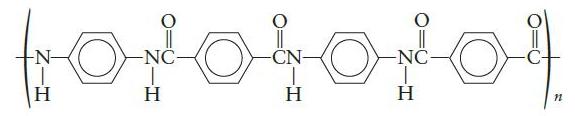
Which monomers are used to make Kevlar?
b. Nomex is a polyaramid used in fire-resistant clothing. It is a copolymer of

torosojot -CN- H H -NC- H -NC- H
Step by Step Solution
3.39 Rating (177 Votes )
There are 3 Steps involved in it
Answer a Kevlar is made from the monomers terephthaloyl chloride and paraphenylenediamine b Nom... View full answer

Get step-by-step solutions from verified subject matter experts


