Predict the potential of each of the following cells: 3+ (a) Cr(s) Cr+ (aq, 0.37 mol-L-)||Pb+ (aq,
Question:
Predict the potential of each of the following cells: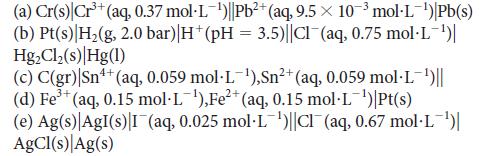
Transcribed Image Text:
3+ (a) Cr(s) Cr³+ (aq, 0.37 mol-L-¹)||Pb²+ (aq, 9.5 10³ mol-L-¹) Pb(s) (b) Pt(s) H₂(g, 2.0 bar) |H* (pH = 3.5)||Cl¯(aq, 0.75 mol·L-¹)| Hg₂Cl₂ (s) Hg(1) (c) C(gr) |Sn¹+ (aq, 0.059 mol-L-¹),Sn²+ (aq, 0.059 mol-L-¹)|| 3+ (d) Fe³+ (aq, 0.15 mol-L-¹), Fe²+ (aq, 0.15 mol-L-¹)|Pt(s) (e) Ag(s) AgI(s) I (aq, 0.025 mol-L¹)||Cl(aq, 0.67 mol.L-¹)| AgCl(s) Ag(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The cell potential can be determined by using the Nernst equation Ecell Ecell 00592n logQ For the ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Water potential in potato cells was determined in the following manner. The initial masses of six groups of potato cores were measured. The potato cores were placed in sucrose solution of various...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
The owners want to do some analysis to see how their sales are doing. Management wants to see how the company did last year and make some decisions about what products to keep along with staffing for...
-
The MLC Corporation produces electronic microwave ovens and uses a perpetual inventory system. Its job costing records yield the following information. Job No. 1 m 2 3 4 *5 6 7 Date Finished Sold...
-
Variance analysis, multiple products. The Detroit Penguins play in the American Ice Hockey League. The Penguins play in the Downtown Arena (owned and managed by the City of Detroit, which has a...
-
Five data entry operators work at the data processing department of the Georgia Bank. Each day for 30 days, the number of defective records in a sample of 250 records typed by these operators has...
-
MARRIOTT: Marketing Research Leads to Expanded Offerings Visit www.pearsonglobaleditions.com/malhotra to read the video case and view the accompanying video. Marriott: Marketing Research Leads to...
-
Frenchvanilla Company earned net income of $ 75,000 during the year ended December 31, 2014. On December 15, Frenchvanilla declared the annual cash dividend on its 5% preferred stock (par value, $...
-
Last year, Dixon Company produced 11,000 units and sold 9,000 units. The company had no beginning inventory Dixon incurred the following costs: Direct materials per unit Direct labor per unit...
-
(a) The pH of 0.015 m HNO 2 (aq) was measured as 2.63. What are the values of K a and pK a of nitrous acid? (b) The pH of 0.10 m C 4 H 9 NH 2 (aq), butylamine, was measured as 12.04. What are the...
-
Calculate the volume of 0.120 m HCl(aq) required to neutralize (a) One-half and (b) All the hydroxide ions in 25.0 mL of 0.412 m KOH(aq). (c) What is the molar concentration of Cl ions at the...
-
The data in the table are from a General Social Survey and concern gender and political party. a. Find the expected counts and report the smallest. Could we use the table as is, without combining...
-
Tanya Tassle owns Born Again Bags, a not for profit business that is registered for GST. Born Again Bags supports the diversion of post-consumer material (waste) into reusable bags to replace plastic...
-
If an organization has an average daily gross patient service revenue $230,000 along with 120 patients preregistered encounters, 150 scheduled encounters, and $100,000 in gross dollars in discharged,...
-
Plan corporation acquired 80% of the outstanding voting stock of Sal Corporation on January 1, 2017 for $14,000,000, when Sals stockholders equity consisted of $8,000,000 capital stock and $1,000,000...
-
Case Illustration Pop Meals is Southeast Asia's fastest growing omnichannel food brand, currently operating through mix model of online delivery, pick-up and app-driven dine-in. For FY2021, Pop...
-
Terras, Inc., leased equipment to Venture Company on January 1, 20X4. The lease is a direct financing lease for a five-year period ending January 1, 20X9. The first annual payments of $1,200,000 was...
-
Who in the factory would be best equipped to provide estimates of the percent completion for work in process?
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
To determine standard cell potentials, measurements are carried out in very dilute solutions rather than at unit activity. Why is this the case?
-
Show that if G o f (H + , aq) = 0 for all T, the potential of the standard hydrogen electrode is zero.
-
How is it possible to deposit Cu on a Au electrode at a potential lower than that corresponding to the reaction Cu 2+ (aq) + 2e Cu(s)?
-
Reading Passage: In the wake of the global health crisis, the number of professionals suffering from workplace burnout has risen and is still rising significantly. Workplace burnout is an ongoing...
-
Prepare an Estimated Tax Worksheet and the payment vouchers for Sean and Sue: Sean D. (SSN 022-00-0947, born 12/12/1952) and Sue P. Summers (SSN 034-00-4362, born 2//9//1955 ) live at 22 Abbot Road,...
-
A random sample of professional wrestlers was obtained, and the annual salary (in dollars) for each was recorded. The summary statistics were x 5 47,500 and n 5 18. Assume the distribution of annual...

Study smarter with the SolutionInn App


