Relative humidity at a particular temperature is defined as The vapor pressure of water at various temperatures
Question:
Relative humidity at a particular temperature is defined as
The vapor pressure of water at various temperatures is given in Table 5A.2.
(a) What is the relative humidity at 30°C when the partial pressure of water is 25.0 Torr?
(b) Explain what would be observed if the temperature of the air were to fall to 25°C.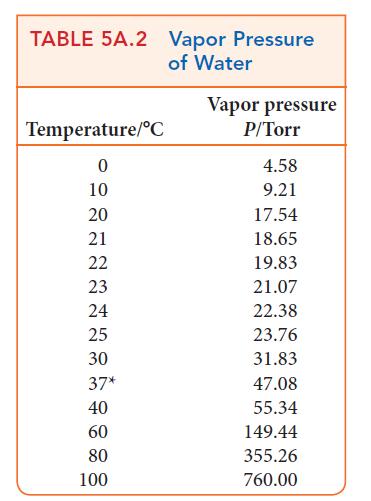
Transcribed Image Text:
Relative humidity = partial pressure of water vapor pressure of water X 100%
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a 785 b Wat...View the full answer

Answered By

Anurag kumar
My experience as a teacher:
Being a teacher is not an easy task. Teaching profession brings with it lots of responsibilities and challenges. But there is nothing wiser than teaching. It is the way of motivating and shaping the character as well as future of a student.
My teaching experience as a chegg teacher is of great fun as well as challenge. Teaching the little ones require huge patience and understanding. Sometimes I used to lose my cool with their excessive naughty behaviors, but I had to be polite as well for not to hurt their innocent mind.
I had to be more loving and caring for they were like pure budding flowers. I don’t know how but in the midst of many challenges I somehow managed to win their love and respect. That is the best gift a teacher can get.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The Dew Point The vapor pressure of water (see Problem 18.88) decreases as the temperature decreases. If the amount of water vapor in the air is kept constant as the air is cooled, a temperature is...
-
The relative humidity of air equals the ratio of the partial pressure of water in the air to the equilibrium vapor pressure of water at the same temperature times 100%. If the relative humidity of...
-
The vapor pressure of water at various temperatures follows: (a) Draw a scatter diagram of these data. What type of relationship seems appropriate in relating y to x? (b) Fit a simple linear...
-
In Exercises 8586, find a. (f g)(x); b. (g f)(x); c. (f g)(3). f(x) = x 2 + 3, g(x) = 4x - 1
-
A 12-V car battery has an internal resistance of 0.4 . (a) What is the current if the battery is shorted momentarily? (b) What is the terminal voltage when the battery delivers a current of 20 A to...
-
Social norms. Social norms often evolve to cope with external costs, without bargaining or law. For example, a social norm in a county in northern California requires that ranchers assume...
-
Consider a randomized pair design with $n$ units where two treatments are randomly assigned to each unit, resulting in a pair of observations $\left(X_{i}, Y_{i} ight)$, for $i=1, \ldots, n$ on each...
-
Northern Communications has the following stockholders equity: Requirements 1. Assuming the preferred stock is cumulative, compute the amount of dividends to preferred stockholders and to common...
-
Suppose that next year you observe a higher equilibrium price but note that the equilibrium quantity of bagels sold is unchanged. What changes to the price of a donut and the price of flour could...
-
8 91426 10 11 (CCM) builds furniture for a range of commercial customers. They build furniture used in hotels and department stores, on cruise liners, ferries and passenger aircraft. One of their...
-
The equilibrium constant K c = 0.56 for the reaction PCl 3 (g) + Cl 2 (g) PCl 5 (g) at 250 C. Upon analysis, 1.50 mol PCl 5 , 3.00 mol PCl 3 , and 0.500 mol Cl 2 were found to be present in a...
-
When sulfuric acid is added to water, so much energy is released as heat that the solution may boil. Would you expect this solution to show a deviation from Raoults law? If so, which kind? Explain...
-
Compare your results in Problems 13.122 and 13.123? Hypertension The data set WALES.DAT contains familial data on blood pressure (bp) in two-communities in South Wales (the Rhondda Fach and the Vale...
-
Randomly selecting a drug screening result and getting one that is a false positive. Randomly selecting a drug screening result and getting one from someone who uses drugs. Determining Whether Events...
-
Generally, new shell entities inserted into existing group structures as part of the corporate reorganization process will meet the definition of business under IFRS 3.
-
Perform the indicated operations, if defined, for the following vectors and matrices. \(\left(\mathbf{B}-\mathbf{v} \mathbf{w}^{T} ight) \mathbf{A}\) -2 1 -3 1 1 A = 1 -3 2 1 32 B = V= W = 0 0 4 5
-
Randomly selecting a drug screening result and getting one that is a false negative. Randomly selecting a drug screening result and getting one from someone who is not a drug user. Determining...
-
If the number of blood donors is n = 20, find the probability that the number with Group O blood is x = 16. Assume that when blood donors are randomly selected, 45% of them have blood that is Group O...
-
A person who receives property as a gift makes no investment to receive the property. Why is a basis assignment to the gift property necessary even though the donee has no investment in the property?
-
g(x) = x 5 5x 6 a. Show that g(x) = 0 has a root, , between x = 1 and x = 2. b. Show that the equation g(x) = 0 can be written as x = (px + q) 1/r , where p, q and r are integers to be found. The...
-
Compound A has molecular formula C 8 H 8 . When treated with excess Br 2 , compound A is converted into compound B, with molecular formula C 8 H 8 Br 2 . Identify the structures of compounds A and B....
-
In some circumstances, dehydrogenation is observed. Dehydrogenation involves the loss of two hydrogen atoms (the reverse of hydrogenation). Analyze each of the following dehydrogenation reactions and...
-
Predict whether each of the following compounds should be aromatic. a. b. c.
-
a) Find the perimeter of a square in which each side measures 13 inches. b) Determine the length of the fence you'd need to enclose a lawn that's a rectangle with sides of 25 ft and 30 ft. c) Find...
-
How can integrative negotiation strategies, rooted in principles of mutual gains and interest-based bargaining, foster collaborative conflict resolution in complex interpersonal or organizational...
-
Oscar, Ltd. is a British subsidiary of an American company. Oscar's functional currency is the pound sterling (). The following exchange rates were in effect during 2021: January 1 1 = ps 1.58 June...

Study smarter with the SolutionInn App


