Predict whether each of the following compounds should be aromatic. a. b. c.
Question:
a.
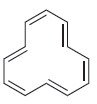
b.
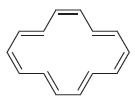
c.
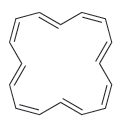
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a No 12 is not a ...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict whether each of the following compounds is molecular or ionic: (a) B2H6 (b) CH3OH (c) LiNO3 (d) Sc2O3 (e) CsBr (f) NOCl (g) NF3 (h) Ag2SO4.
-
Using solubility guidelines, predict whether each of the following compounds is soluble or insoluble in water: (a) MgBr2 (b) PbI2 (c) (NH4)2CO3 (d) Sr(OH)2 (e) ZnSO4.
-
Predict whether each of the following compounds is soluble in water: (a) AgI (b) Na2CO3 (c) BaCl2 (d) Al(OH)3 (e) Zn(CH3COO)2.
-
Cinderella's income increases by 25%. She decides to increase her purchases of glass slippers by 40%. To her, glass slippers are a(n)____________good and her income elasticity of demand for glass...
-
The results of a sieve test below give the percentage passing through the sieve. (a) Using a spreadsheet, plot the particle-size distribution. (b) Calculate the uniformity coefficient. (c) Calculate...
-
Ryanair Holdings PLC (LSE: RYA) operates a low-fare scheduled passenger airline serving short-haul, point-to-point routes between Ireland, the United Kingdom, Continental Europe, and Morocco. The...
-
Visit www.pearsonglobaleditions.com/malhotra to read the video case and view the accompanying video. Marriott: Marketing Research Leads to Expanded Offerings highlights Marriott's discovery from...
-
The following events apply to R&L Logging Company for the 2010 fiscal year. 1. The company started when it acquired $80,000 cash from the issue of common stock. 2. Purchased a new skidder that cost...
-
Carla Vista Inc. has beginning work in process $30,300, direct materials used $283,200, direct labor $259.600, total manufacturing overhead $212,400, and ending work in process $37,000. What are the...
-
NUNA MUSIC Tiral Balances February 28 3014 Unadjusted Trial Adjusted Trial Balance Adjustments Balance Account Dr Cr Dr Cr Dr Cr Cash 14000 14000 Accounts receivable 32000 31000 63000 Prepaid...
-
In some circumstances, dehydrogenation is observed. Dehydrogenation involves the loss of two hydrogen atoms (the reverse of hydrogenation). Analyze each of the following dehydrogenation reactions and...
-
The cyclopropenyl cation has a three-membered ring that contains a continuous system of overlapping p orbitals. This system contains a total of two Ï electrons. Using a Frost circle, draw an...
-
Which two elements would you expect to exhibit the greatest similarity in physical and chemical properties: B, C, Hf, Pb, Pr, Sn? Explain your choice.
-
Mike's gross yearly income is $ 6 6 , 9 9 0 . He is single with no children, and his tax deductible purchases are: Charitable contributions: $ 2 , 2 4 9 Mortgage Interest: $ 2 , 1 0 1 Student Loans:...
-
Save Co. had the following information for its fiscal year ending December 31, 2023: Net income for 2023 $520,000 Retained earnings, January 1, 2023 210,000 Cash dividends declared on common shares...
-
Pawn Company makes 6,248 chess sets per year. It is debating on whether it should continue making the chess sets or buy them from an outside suppler for $22 per unit. Below is information regarding...
-
Shown below is an adjusted trial balance for Cornell Products, Incorporated, on December 31: Cornell Products, Incorporated Adjusted Trial Balance December 31, Year 1 Cash Accounts Receivable Office...
-
Joe's Tax Service provides professional accounting and tax services. All tax return services and classified into one of three categories: standard, multiform, and complex. Following is information...
-
What is the best way to really prove to yourself that you understand an idea?
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
Assign R or S configuration to each chirality center in the followingmolecules: (b) NH2 (a) . "C r "O
-
Draw tetrahedral representations of the following molecules: (a) The 2S, 3R enantiomer of 2, 3-dibromopentarie (b) The meso form of 3,5-heptanediol H. 4-Methyl-2-pentanone 3-Methylpentanal
-
Draw the meso form of each of the following molecules, and indicate the plane of symmetry in each: (b) (c) . (a) CH H,H,H,, - CHCH2CH2CHH CH
-
Imagine yourself at a fair playing one of the midway games. Pick a game and calculate the expected value and post your results along with how you calculated them.
-
1. What is the ratio of 2 lengths which are 25 cm and 7.5 m respectively?
-
If f(x, t) does not depend on x, then we have x' = f(t), so that x(t) = (0 to t) f(u)du. Show that, in this case, the RK4 algorithm reduces to Simpson's rule.

Study smarter with the SolutionInn App


