Select from each of the following pairs of Lewis structures the one that is likely to make
Question:
Select from each of the following pairs of Lewis structures the one that is likely to make the dominant contribution to a resonance hybrid. Explain your selection.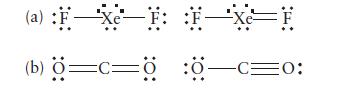
Transcribed Image Text:
(a) :FXeF: :F—Xe=F (b) Ö=C=0 :0-C=0: O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a The first structure is ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Select from each of the following pairs of Lewis structures the one that is likely to make the dominant contribution to a resonance hybrid. Explain your selection. (a) N=N=0 :N=N0: 73- :: 73- :: ::...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Select the compound in each of the following pairs that will be converted to the corresponding alkyl bromide more rapidly on being treated with hydrogen bromide. Explain the reason for your choice....
-
DrinkOh Limited uses an application service provider to process its payroll. Its employees enter their hours using their smart phones. The payroll clerk collects the smart phone data and transmits it...
-
Which type of cost system, process or job order, would be best suited for each of the following: (a) TV assembler, (b) Building contractor, (c) Automobile repair shop, (d) Paper manufacturer, (e)...
-
Solve the given inequalities. Graph each solution. It is suggested that you also graph the function on a calculator as a check. X x + 3 > 1
-
If two charged particles \(M\) and \(S\) are at rest relative to each other, there is no magnetic force between them. Suppose instead that particle \(M\) is moving relative to particle \(S\) while...
-
Frank Dorrance, a senior audit manager for Bright and Lorren, CPAs, has recently been informed that the firm plans to promote him to partner within the next year or two if he continues to perform at...
-
Which one of the metal atoms whose electronic structure given below forms a nitrate of the type M(NO3)2? 37 A. 2.8.0 B. 2.8.1 C. 2.8.2 D. 2.8.3
-
Predict the number of valence electrons present for each of the following ions: (a) Mn 4+ ; (b) Rh 3+ ; (c) Co 3+ ; (d) P 3+ .
-
Predict the bond angles at the central atom of the following molecules and ions: (a) Ozone, O 3 ; (b) Azide ion, N 3 ; (c) Cyanate ion, CNO 2 ; (d) Hydronium ion, H 3 O + .
-
John Holland Incorporated provides you with the following information. The company does not report any book tax differences and is subject to a 21% income tax rate. Holland is a US Corporate tax...
-
In the hierarchical data model, the mapping from parent to child is a. 1:1 (one-to-one). b. 1:N (one-to-many). c. N:N (many-to-many). d. N:1 (many-to-one).
-
Consider Bobs Blu-ray company described in Problem 3. Assume that Blu-ray production is a perfectly competitive industry. For each of the following questions, explain your answers. a. What is Bobs...
-
How will the following events affect the demand for money? In each case, specify whether there is a shift of the demand curve or a movement along the demand curve and its direction. a. There is a...
-
Although the U.S. Federal Reserve doesnt use changes in reserve requirements to manage the money supply, the central bank of Albernia does. The commercial banks of Albernia have $100 million in...
-
The market for toys during the holiday season is intensely competitive. Before the 2018 holiday season, Toys R Us and the Bon-Ton department store chain had closed, and Sears had closed many of its...
-
True or False: To make a natural monopolist behave more efficiently, subsidies will work better than price controls.
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Draw all constitutional isomers with molecular formula C 3 H 8 O, and rank them in terms of increasing acidity.
-
Consider the structure of cyclopentadiene and then answer the following questions: (a) How many sp 3 -hybridized carbon atoms are present in the structure of cyclopentadiene? (b) Identify the most...
-
When (1R, 2R)-2-bromocyclohexanol is treated with a strong base, an epoxide (cyclic ether) is formed. Suggest a mechanism for formation of the epoxide: Strong base Br An epoxide
-
Ivanhoe Company reports the following operating results for the month of April. IVANHOECOMPANY CVP Income Statement For the Month Ended April 30, 2022 Total Per Unit Percent of Sales Sales (4,860...
-
How do you manage dependencies and relationships between datasets, ensuring that interrelated data elements are migrated cohesively to maintain data integrity and relational integrity?
-
Given the sequence a defined recursively as follows: a = 0 akak-1+4k - 4 when k>1

Study smarter with the SolutionInn App


