Select from each of the following pairs of Lewis structures the one that is likely to make
Question:
Select from each of the following pairs of Lewis structures the one that is likely to make the dominant contribution to a resonance hybrid. Explain your selection.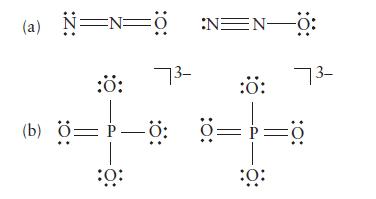
Transcribed Image Text:
(a) N=N=0 :N=N0: 73- :Ö: 73- :Ö: :Ö: (b) Ö=P—ö Ö==d :0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The concept of resonance in chemistry refers to the delocalization of electrons within a molecule that has conjugated bonds In other words it describe...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Select from each of the following pairs of Lewis structures the one that is likely to make the dominant contribution to a resonance hybrid. Explain your selection. (a) :FXeF: :FXe=F (b) =C=0 :0-C=0:...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Select the compound in each of the following pairs that will be converted to the corresponding alkyl bromide more rapidly on being treated with hydrogen bromide. Explain the reason for your choice....
-
how good are your with western civilization history?? choose one of the following topics topic 1: based on the texts by kafka and eliot, (writing on one or the other or both), discuss how the writers...
-
Pocket Pilot Inc. is considering an investment in new equipment that will be used to manufacture a mobile communications device. The device is expected to generate additional annual sales of 6,000...
-
Use integration by substitution and the Fundamental Theorem to evaluate the definite integrals in Problems. x x dx
-
Based on the information for Norwegian Bristling Company for the year ended December 31, 2018: a. How much is the firms net working capital and what is the debt ratio? b. Complete a statement of cash...
-
Humble Management Services opens for business and completes these transactions in September. Sept. 1 Henry Humble, the owner, invests $38,000 cash along with office equipment valued at $15,000 in the...
-
Question Two. Evaluate the levels of management Question Three. [15 marks] A chief executive officer cannot perform all the activities in organization; discuss the importance of the organizing...
-
Predict the number of valence electrons present for each of the following ions: (a) In + ; (b) Tc 2 + ; (c) Ta 2 + ; (d) Re + .
-
Predict the bond angles at the central atom of the following molecules and ions: (a) OF 2 ; (b) ClO 2 ; (c) NO 2 ; (d) SeCl 2 .
-
Design a relational database corresponding to the E-R diagram of Figure. model year address Ticense driver-id name location car owns report-number uOSAad date accident participated driver...
-
What role will the compensation design play in motivating the new sales representative?
-
What are the sales objectives for the new sales representative?
-
Explain what the market pay line is. How is it used in the context of pay level policies such as market lead, market lag, and market match?
-
What are some potential disadvantages of the HSA option?
-
What are some advantages of implementing the HSA option?
-
Hortense earns a wage of $10 per hour and chooses to work 35 hours per week. One day, her employer tells her that while he will continue to pay her $10 an hour for her first 35 hours each week, he...
-
Suppose you are comparing just two means. Among the possible statistics you could use is the difference in means, the MAD, or the max min (the difference between the largest mean and the smallest...
-
Draw both chair conformations for each of the following compounds. In each case, identify the more stable chair conformation: (a) Methylcyclohexane (b) Trans-1,2-Diisopropylcyclohexane (c)...
-
Identify the product of the following reaction: Br Br ONa 2 NaBr C4H3O2 Nao
-
When trans-1-phenylpropene is treated with bromine, some syn addition is observed. Explain why the presence of a phenyl group causes a loss of stereospecificity. Br + En Br2 En Br Br Syn addition...
-
What is a good approach to measure a mass very precisely to see if its weight appears to fluctuate over time Explain.
-
The walls in your home have 25 mm of expanded polystyrene and 25 mm of rigid urethane foam. What is the R-value of your insulation? Check the R-value of 25 mm of expanded polystyrene. Check the...
-
Year Cash Flows 0 1 2 3 4 Project -300 250 150 70 350 A Project -300 200 100 100 400 B Assuming a discount rate of 10% Which project would you choose to invest in and why? Use calculations to support...

Study smarter with the SolutionInn App


