There are three isomers of dichlorobenzene, C 6 H 4 Cl 2 , which differ in the
Question:
There are three isomers of dichlorobenzene, C6H4Cl2, which differ in the relative positions of the chlorine atoms on the benzene ring.
(a) Which of the three forms are polar?
(b) Which has the largest dipole moment?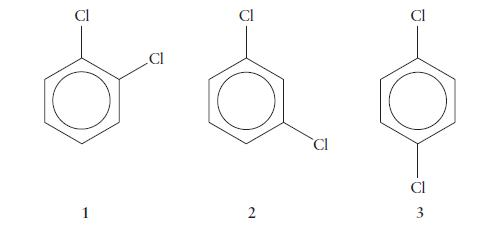
Transcribed Image Text:
1 CI 2 Cl Cl 1. 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a 1 and 2 are ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
There are three isomers of dichlorobenzene, one of which has now replaced naphthalene as the main constituent of mothballs. a. Identify the ortho, the meta, and the para isomers of dichlorobenzene....
-
There are three isomers of difluoroethene, C 2 H 2 F 2 , which differ in the locations of the fluorine atoms. (a) Which of the forms are polar? (b) Which has the largest dipole moment? F F C= 1 C O H...
-
Which one of the dichlorobenzene isomers does not have a dipole moment? Which one has the largest dipole moment? Compare your answers with the dipole moments calculated using the molecular-modeling...
-
TCP: the client sends only 1 message to the server hello from TCP Client and the server responds with the uppercase message. Update the program / make a simple chat program so that The client can...
-
In what major respect would budget performance reports prepared for the use of plant managers of a manufacturing business with cost centers differ from those prepared for the use of the various...
-
Briefly describe the role an online information service could play in a used car purchase transaction.
-
Discuss the financing indicators used to determine the functional currency of an entity?
-
Use the Rolling Hills data from Problem 14-27A. Requirements 1. Prepare the 2012 statement of cash flows by the direct method. 2. How will what you learned in this problem help you evaluate an...
-
(a) The Treasury desk of a global bank incorporated in UK wants to invest GBP 200 million on 1st January, 2019 for a period of 6 months and has the following options: (1) The Equity Trading desk in...
-
An organic compound distilled from wood was found to have a molar mass of 32.04 g mol 1 and the following composition by mass: 37.5% C, 12.6% H, and 49.9% O. (a) Write the Lewis structure of the...
-
Predict which of the following pairs of ions would have the greatest coulombic attraction in a solid compound: (a) Mg 2+ , S 2+ ; (b) Mg 2+ , Se 2+ ; (c) Mg 2+ , O 2+ .
-
How is the order of a differential equation determined?
-
Cavendish plc is a Stock Exchange listed business whose main activity is producing a low-value material used in large quantities in the building/construction industries. Production is such that a...
-
Antithesis Ltd sells its service on credit at the rate of 6 million a year. Customers take varying lengths of time to pay, but the average is 65 days. The business does not experience any significant...
-
You have overheard the following statement: If an investor holds shares in about 20 different businesses all of the risk is eliminated and the portfolio will give a return equal to the risk-free...
-
Shah plc has a cost of equity of 17 per cent p.a. The business is expected to pay a dividend in one years time of 0.27 per share. Dividends are expected to grow at a steady 5 per cent each year for...
-
XYZ Ltd, an unlisted business, is quite like ABC plc, a listed one, in term of activities, gearing and dividend policy. XYZ Ltd recently paid a dividend for the year of 0.20 per share. ABC plcs...
-
True or False: When the interest rate goes up, investment becomes more desirable.
-
How much more interest will be earned if $5000 is invested for 6 years at 7% compounded continuously, instead of at 7% compounded quarterly?
-
The reaction \[\mathrm{A} \longrightarrow \mathrm{B}+\mathrm{C}\] is known to be zero order in \(\mathrm{A}\) and to have a rate constant of \(5.0 \times 10^{-2} \mathrm{~mol} \mathrm{~L}^{-1}...
-
The reaction \[2 \mathrm{I}^{-}(a q)+\mathrm{S}_{2} \mathrm{O}_{8}{ }^{2-}(a q) \longrightarrow \mathrm{I}_{2}(a q)+2 \mathrm{SO}_{4}{ }^{2-}(a q)\] was studied at \(25^{\circ} \mathrm{C}\). The...
-
The reaction \[2 \mathrm{NO}(g)+\mathrm{Cl}_{2}(g) \longrightarrow 2 \mathrm{NOCl}(g)\] was studied at \(-10^{\circ} \mathrm{C}\). The following results were obtained, where \[\text { Rate...
-
Show transcribed image text Hofburg's standard quantities for 1 unit of product include 2 pounds of materials and 1.5 labor hours. The standard rates are $2 per pound and $7 per hour. The standard...
-
A hotel in Orlando, Florida, experiences peak periods and slower times. How should prices be adjusted during peak periods? During slow times? Why? Clark Manufacturing offers two product lines, IN2...
-
Sophie Chan owns 100,000 shares of PAT Company. PAT is selling for 40 per share, so her investment is worth 4,000,000. Chan reinvests the gross amount of all dividends received to purchase additional...

Study smarter with the SolutionInn App


