To analyze the alcohol content of a certain wine, a chemist needs 1.00 L of an aqueous
Question:
To analyze the alcohol content of a certain wine, a chemist needs 1.00 L of an aqueous 0.200 M K2Cr2O7 (potassium dichromate) solution. How much solid K2Cr2O7 must be weighed out to make this solution?
Figure 4.9
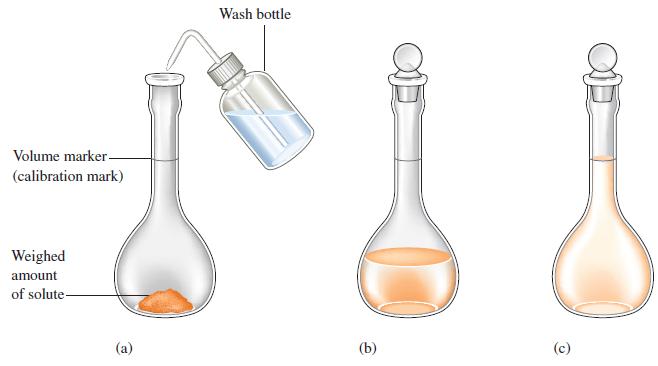
Transcribed Image Text:
Volume marker- (calibration mark) Weighed amount of solute- (a) Wash bottle (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
First determine the moles of KCrO7 required 0200 mol KCrO7 L solution 100 L ...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
The energy content of a certain food is to be determined in a bomb calorimeter that contains 3 kg of water by burning a 2-g sample of it in the presence of 100 g of air in the reaction chamber. If...
-
What is the is the effect of abolition of an import quota on (i) national saving, (ii) domestic investment, (iii) NCO, (iv) the real exchange rate, and (v) net exports?
-
A rod is bent into a circular arc of radius 4 in. as shown. For the given loading, determine the internal forces at point J when 30 θ = °. 12 Ih B. 4 in.
-
A large restaurant chain (see Example 1.6) periodically offers special milk shake flavors for a limited time. Suppose that the contenders for the next special flavor are Green Mint, Orange Crush, Egg...
-
Apply the marketing-myopia concept to print media, magazines, and newspapers. What is the implication?
-
TufStuff, Inc., sells a wide range of drums, bins, boxes, and other containers that are used in the chemical industry. One of the companys products is a heavy-duty corrosion-resistant metal drum,...
-
An aluminum flagpole is 34 m high. By how much does its length increase as the temperature increases by 23 C? (The linear expansion coefficient of aluminum is 23 x 10-6 /C)
-
Using the solubility rules in Table 4.1, predict what will happen when the following pairs of solutions are mixed. a. KNO 3 (aq) and BaCl 2 (aq) b. Na 2 SO 4 (aq) and Pb(NO 3 )2(aq) c. KOH(aq) and...
-
Typical blood serum is about 0.14 M NaCl. What volume of blood contains 1.0 mg of NaCl?
-
In problem the given function is analytic at a = 0. Use appropriate series in (2) and long division to find the first four nonzero terms of the Maclaurin series of the given function.
-
Net manufacturing income $50,000 Interest on overdue receivables 4,000 Interest on investments 2,000 Net income for tax purposes 56,000 Less: Charitable donations (1,000) Non-capital losses from...
-
Do some of the criteria give different investment recommendations? Why? The acceptance of the project proposal is dependent on four different factors, referred to as "performance hurdles" in the...
-
What are the fundamental characteristic of fund accounting for nonprofits? Explain briefly
-
What are the accounting principle guides that recognition of contributions and grants in financial statements?
-
What are the levels of risk control? Explain the application of each of the controls from the hierarchy of control measures.
-
Marsh Products, Inc., reported the following on the company's income statement in 2012 and 2011: a. Determine the number of times interest charges were earned for 2011 and 2012. Round to one decimal...
-
on 8 For the following set of lengths 130, 170, 160, 160, 150, 190 Third quartile is: et red d out of Select one: O a. 160 a question O b. 145 O c. 175 O d. 180
-
What is supersaturation? Under what conditions is it possible to supersaturate a solution? What is the metastable region?
-
Does the commonly reported solubility of an inorganic compound in water pertain to large crystals or small crystals? Why?
-
Can an inorganic compound have more than one form of hydrate?
-
Discuss the role of linear programming in production planning under uncertainty and how it can be used to handle demand fluctuations.
-
1. Provide an example from everyday life of positive reinforcement, negative reinforcement, positive punishment, and negative punishment. 2. Describe the US, UR, CS, and CR from the Pavlov's dog...
-
Suppose you are going to receive $13,500 per year for five years. The appropriate interest rate is 6.8%. a-1 What is the present value of the payments if they are in the form of an ordinary annuity?...

Study smarter with the SolutionInn App


