Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength:
Question:
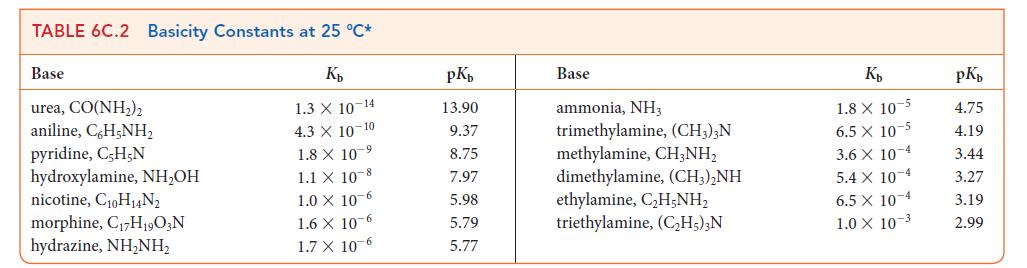
Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength: C10H14N2 (nicotine), ClO2, (CH3)3N, HSO3–.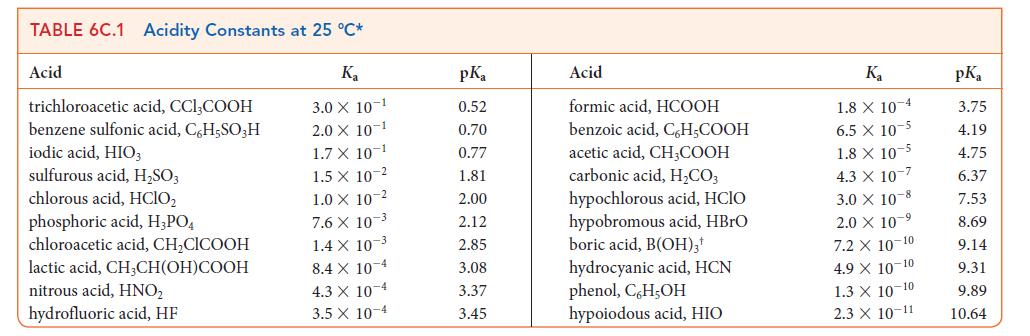
Transcribed Image Text:
TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X 10-1 2.0 X 10-1 1.7 X 10-1 1.5 X 1.0 X 10-2 Acid trichloroacetic acid, CCI,COOH benzene sulfonic acid, C6H5SO3H iodic acid, HIO3 sulfurous acid, HSO3 chlorous acid, HClO phosphoric acid, H3PO4 chloroacetic acid, CHCICOOH lactic acid, CHCH(OH)COOH nitrous acid, HNO hydrofluoric acid, HF 10 2 7.6 x 10-3 1.4 X 10-3 8.4 X 10 4 4.3 X 10-4 3.5 x 10-4 pKa 0.52 0.70 0.77 1.81 2.00 2.12 2.85 3.08 3.37 3.45 Acid formic acid, HCOOH benzoic acid, C,HCOOH acetic acid, CH,COOH carbonic acid, HCO3 hypochlorous acid, HCIO hypobromous acid, HBrO boric acid, B(OH)3 hydrocyanic acid, HCN phenol, C,H,OH hypoiodous acid, HIO K 1.8 X 10 4 6.5 X 10-5 1.8 X 10-5 4.3 X 107 3.0 X 108 2.0 10- 7.2 X 10-10 4.9 X 10-10 1.3 X 10-10 2.3 X 10-11 pKa 3.75 4.19 4.75 6.37 7.53 8.69 9.14 9.31 9.89 10.64
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Decreasing pK will correspond to in...View the full answer

Answered By

Daniel Kimutai
I am a competent academic expert who delivers excellent writing content from various subjects that pertain to academics. It includes Electronics engineering, History, Economics, Government, Management, IT, Religion, English, Psychology, Sociology, among others. By using Grammarly and Turnitin tools, I make sure that the writing content is original and delivered in time. For seven years, I have worked as a freelance writer, and many scholars have achieved their career dreams through my assistance.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength: F , NH 3 , CH 3 CO 2 , C 5 H 5 N (pyridine). TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X 10-1 2.0...
-
Arrange the following bases in order of increasing strength on the basis of the pK a values of their conjugate acids, which are given in parentheses: (a) Aniline (4.63; see Exercise 6C.12); (b)...
-
Arrange the following bases in order of increasing strength on the basis of the pK a values of their conjugate acids, which are given in parentheses: (a) Ammonia (9.26); (b) Methylamine (10.56); (c)...
-
Allocation of Common Costs. The cities of Albany, Troy and Schenectady are considering the implementation of a new program to handle disposal of hazardous waste to comply with a new, more stringent...
-
Daily high temperatures in St. Louis for the last week were as follows: 93, 94, 93, 95, 96, 88, 90 (yesterday). (a) Forecast the high temperature today, using a 3-day moving average. (b) Forecast the...
-
Visit Trackur.com (www.trackur.com) and read about the services Trackur offers. Some analysts have referred to the practice of mining social media as being similar to conducting a focus group. Is...
-
Statement of cash flows; investing and financing activities Refer to the situation described in BE 4-13. Prepare the cash flows from investing and financing activities sections of HHC's statement of...
-
How can systems theory inform the development of holistic frameworks for assessing the multifaceted dimensions of empowerment within complex adaptive systems ? Explain
-
Calculate the molar concentrations of H 2 SO 3 , HSO 3 , SO 3 2 , H 3 O + , and OH present in 0.125 m H 2 SO 3 (aq).
-
Calculate the molar solubility of each of the following sparingly soluble compounds in its respective solution: iron(III) hydroxide at (a) pH = 11.0; (b) pH = 3.0; iron(II) hydroxide at (c) pH = 8.0;...
-
Complete the accompanying run ticket and give the entry to record the sale of the oil at $75/bbl assuming a severance tax rate of 5% and a 1/5 RI. The operator is Montgomery Oil Company and the run...
-
A fluid bed incinerator, $3 \mathrm{~m}$ in diameter and $0.56 \mathrm{~m}$ high, operates at $850^{\circ} \mathrm{C}$ using a sand bed. The sand density is $2.5 \mathrm{~g} / \mathrm{cm}^{3}$, and...
-
A solution of \(13.5 \%\) by weight of polyisoprene has the following power-law parameters: \(\Lambda=5000 \mathrm{~Pa} \cdot \mathrm{s}^{n}\) and \(n=0.2\). Consider the flow of such a solution in a...
-
A mixture of titanium $(\mathrm{SG}=4.5)$ and silica $(\mathrm{SG}=2.65)$ particles, with diameters ranging from 50 to $300 \mu \mathrm{m}$, is dropped into a tank in which water is flowing upward....
-
After blowing up a balloon, you release it without tying off the opening, and it flies out of your hand. If the diameter of the balloon is 6 in., the pressure inside it is $1 \mathrm{psig}$, and the...
-
A speedboat is propelled by a water jet motor that takes water in at the bow through a $10 \mathrm{~cm}$ diameter duct and discharges it through a $50 \mathrm{~mm}$ diameter nozzle at a rate of $80...
-
Guess Company manufactures a variety of natural fabrics for the clothing industry. The following cost data are available for the month of January. There were 80,000 units in process on January 1...
-
A firm has the following balance sheet: Assets Cash Accounts receivable Inventory Plant and equipment $ 15,000 150,000 92,000 170,000 $427,000 Liabilities and Equity Accounts payable Long-term debt...
-
At 39.9C, a solution of ethanol (x 1 = 0.9006, P * 1 = 130.4 Torr) and isooctane (P * 2 = 43.9 Torr) forms a vapor phase with y 1 = 0.6667 at a total pressure of 185.9 Torr. a. Calculate the activity...
-
Calculate the solubility of H 2 S in 1 L of water if its pressure above the solution is 2.75 Pa. The density of water at this temperature is 997kg m 3 .
-
The binding of NADH to human liver mitochondrial isozyme was studied [Biochemistry 28 (1989): 5367] and it was determined that only a single binding site is present with K = 2.0 10 7 M 1 . What...
-
For the frame shown below, assume that all members are axially rigid with the following properties: E = 29,000 ksi, and I = 400 in.4 . The frame is subjected to a vertical load of 30 kips and at the...
-
The bridge has a continuous girder and a truss. All truss members have the same cross-sectional area of 2 in.2 and can deform axially. Assume that the girder is axially rigid with the moment of...
-
Why is it advantageous for candidates to stay in contact with prospective employers after an interview? Provide at least three reasons please with more details explanation and recent references

Study smarter with the SolutionInn App


