Using Table 7.2, arrange the following species according to their strength as bases: H 2 O, F
Question:
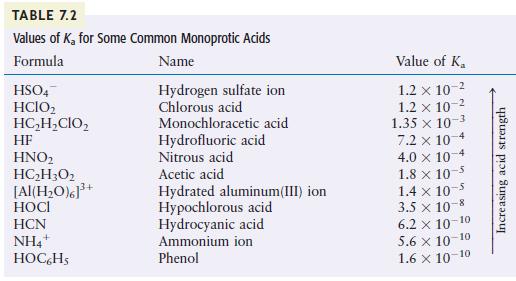
Using Table 7.2, arrange the following species according to their strength as bases: H2O, F–, Cl–, NO2–, and CN–.
Transcribed Image Text:
TABLE 7.2 Values of K for Some Common Monoprotic Acids Formula Name Hydrogen sulfate ion Chlorous acid HSO4 HCIO HCHCIO HF HNO HCH302 [Al(HO)]+ HCN NH4 HOCHS + 3+ Monochloracetic acid Hydrofluoric acid Nitrous acid Acetic acid Hydrated aluminum(III) ion Hypochlorous acid Hydrocyanic acid Ammonium ion Phenol Value of K 1.2 x 10- 1.2 x 10- 1.35 x 10-3 7.2 x 10 4.0 x 101 -4 1.8 x 10 1.4 x 10-5 3.5 10-8 6.2 10-10 5.6 10-10 1.6 10-10 Increasing acid strength
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Remember that water is a stronger base than th...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(A) Refer only to the periodic table on the inside front cover, and arrange the following species in order of increasing size: Ti 2+ , V 3+ , Ca 2+ , Br - , and Sr 2+ . (B) Refer only to the periodic...
-
Compare the following two regressions: i. Y, =B + B, X, + e, ii. Y, =B + B(2X) + e, Equation i. is exactly the regression we've been working with thus far, so all the formulas we've derived thus far...
-
Arrange the following species in order of increasing oxidation number of the sulfur atom: (a) H2S, (b) S8 (c) H2SO4, (d) S2-, (e) HS-, (f) SO2, (g) SO3.
-
Besides warehouse layout decisions, what are some other applications where ranking items according to bang/buck might make sense?
-
Light with wavelength = 0.50m falls on a slit of width b = 10m at an angle 0 = 30 to its normal. Find the angular position of the first minima located on both sides of the central Fraunhofer maximum.
-
True or false: When an OSPF route sends its link state information, it is sent only to those nodes directly attached neighbors. Explain.
-
In February 2014, defendant Ibrahim M. Shihadeh, d/b/a Creative Designs Kitchen and Baths, agreed to purchase 25% of his anticipated natural gas needs at a fixed price for the 201415 and 201516...
-
Zoom-o-licious makes candy bars for vending machines and sells them to vendors in cases of 30 bars. Although Zoom-o-licious makes a variety of candy, the cost differences are insignificant, and the...
-
Explain why there are the same number of tables in the relational model as compared to the number of entities in the logical model.
-
At 60C the value of K w is 1 10 13 . a. Using Le Chteliers principle, predict whether the reaction is exothermic (releases energy) or endothermic (absorbs energy). b. Calculate [H + ] and [OH ] in...
-
A type of reaction we will study is that having a very small \(K\) value \((K < <1)\). Solving for equilibrium concentrations in an equilibrium problem usually requires many mathematical operations...
-
The ledger of Danieal Rental Agency on March 31 of the current year includes the selected accounts, shown on the next page, before adjusting entries have been prepared. An analysis of the accounts...
-
Research a controversial event or legal case raising issues of media ethics or media law from the last ten years. "Specific media ethics or law issue" should be interpreted broadly. You may select...
-
Explain why an adversarial relationship exists between the media and law enforcement.?
-
How does the media portray law enforcement in todays society? explain briefly
-
Give a Christian worldview perspective on social media and law enforcement ?
-
Under the FTC (Federal Trade Commission)the government enforces laws regarding false advertisements. These advertisements are designed to persuade the public to action. Why are these studied in mass...
-
Assuming that the clerk had required a cosigner based on Sages race or gender, what act prohibits such credit discrimination? Leota Sage saw a local motorcycle dealers newspaper advertisement for a...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
(a) Calculate the work associated with the isothermal, reversible expansion of 1.000 mol of ideal gas molecules from 7.00 L to 15.50 L at 25.0 C. (b)Calculate the work associated with the...
-
Neutralization reactions occurring when acids and bases are mixed can be very exothermic. Suppose you are investigating how the heat released in various neutralization reactions is related to the...
-
On the basis of the structures of each of the following molecules, predict which ones would be most likely to have a residual entropy in their crystal forms at T = 0: (a) CO 2 ; (b) NO; (c) N 2 O;...
-
profile each of our main competitors, specifically, what they are doing to market their bananas. Highlight any brand extensions you may find and group the competitors' activities by country....
-
A online radio platform conducted an experiment.In the treatment condition, a playlist with customized new music suggestions was featured on the subscribers' pages (vs. no playlist in the control...
-
Briefly describe the program, and the rewards. 2.How does this loyalty program target the best customers? 3.How is it "sticky" to prevent you from switching? (hint, if you are also with a...

Study smarter with the SolutionInn App


