What is (a) The electrolyte and (b) The oxidizing agent in the mercury cell shown here? (c)
Question:
What is
(a) The electrolyte and
(b) The oxidizing agent in the mercury cell shown here?
(c) Write the overall cell reaction for a mercury cell.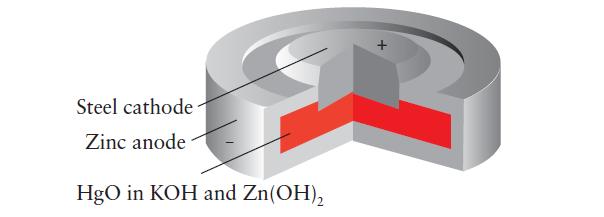
Transcribed Image Text:
Steel cathode Zinc anode HgO in KOH and Zn(OH)₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a KOHaqH...View the full answer

Answered By

Sandhya Sharma
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
119+ Reviews
214+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
For the alkaline Leclanch cell. (a) write the overall cell reaction. (b) Determine E cell for that cell reaction. Figure 19.14 The Leclanch (Dry) Cell The most common form of voltaic cell is the...
-
From the Nerst equation E = E0 + RT/nF ln a... show that for a pH electrode that an uncertainty in the measurement of the potential leads to an error of 4% in the measurement of the pH.
-
The following half reactions play important roles in metabolism. 1/2 O2 1 2H+ + 2e H2O NADH + H+ NAD+ + 2H+ + 2e- Which of these two is a half reaction of oxidation? Which one is a half reaction of...
-
Show that 1. Every differentiable concave function is pseudoconcave. 2. Every pseudoconcave function is quasiconcave
-
The Pacific Corporation operates car rental agencies at more than 20 airports. Customers can choose from one of three contracts for car rentals of one day or less: Contract 1: $50 for the day...
-
The adjusted trial balance of Pacific Scientific Corporation on December 31, 2011, the end of the company's fiscal year, contained the following income statement items ($ in millions): sales revenue,...
-
An indirect way of measuring of secondary emission from ponds or large bodies of water used in waste treatment is to measure the concentration and velocity over the surface. The data can then be...
-
Graphical derivation and interpreting beta You are analyzing the performance of two stocks. The first, shown in Panel A, is Cyclical Industries Incorporated. Cyclical Industries makes machine tools...
-
Q. Today, social, mobile, and local marketing are the fastest-growing forms of online marketing. This assignment is to plan your own business. A. Describe its business model mentioning the comparison...
-
(a) Why are leadantimony grids used as electrodes in the leadacid battery rather than smooth plates? (b) What is the reducing agent in the leadacid battery? (c) The leadacid cell potential is about 2...
-
(a) Write the cell reaction for the leadacid battery. (b) Explain how each of the following change in a leadacid battery during discharge: pH; amount of PbO 2 ; total amount of lead in the battery.
-
Locate data on the size of the international economy, including data on international trade, foreign direct investment by U.S. firms, and investment in the United States by foreign firms. Useful web...
-
If you work in a healthcare organization, do you know how your finance operation is organized? Please describe.
-
What is needed in order to make a determination that work had been accomplished?
-
What do the planned value for the work elements add up to? How is it measured?
-
What is the essential part of creating a plan for the project in EVM?
-
What is a cost variance?
-
Identify the accounts in which each of the following transactions for Doms Furniture, a custom manufacturer of oak tables and chairs, would be debited and credited: 1. Issued oak materials into...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Propose an efficient synthesis for each of the following transformations: (a) (b)
-
The compound below is believed to be a wasp pheromone. Draw the major product formed when this compound is hydrolyzed in aqueous acid:
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OMe Meo Meo Br Br
-
Bob wants to retire. He knows how much he'll receive for his monthly pension. What will help him feel secure that inflation won't increase his bills so much that he can't afford to pay them?
-
Youhaveidentifiedfourstocksthatmaybeunderpriced.Asaresult,youcollectedthefollowinginformation. Inaddition,theexpectedmarketreturn,themarketvolatility,andtherisk-freerateareassumedtobe11%,20%,and 3%,...
-
You estimate that Beta for Infosys as 1.3 using the Indian stock index as the benchmark. If you had used the MSCI world index you would estimate Beta for Infosys to be 0.8. If the Beta between Indian...

Study smarter with the SolutionInn App


