Write the half-reactions and the balanced equation for the cell reaction for each of the following galvanic
Question:
Write the half-reactions and the balanced equation for the cell reaction for each of the following galvanic cells: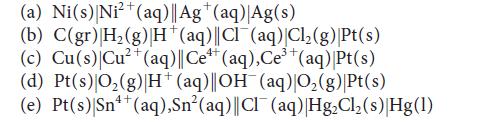
Transcribed Image Text:
(a) Ni(s) Ni²+ (aq)|| Ag+(aq) Ag(s) (b) C(gr) |H₂(g) H*(aq)||Cl¯(aq)|Cl₂(g)|Pt(s) (c) Cu(s) Cu²+ (aq)||Ce+ (aq), Ce³+ (aq) |Pt(s) (d) Pt(s)|O₂(g) |H+ (aq)||OH(aq)|O₂(g)|Pt(s) (e) Pt(s)|Sn++ (aq),Sn²(aq)||Cl¯ (aq) |Hg₂Cl₂(s) Hg(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a anode Nis Ni aq 2 e cathode Ag aq e Ags overall 2 Ag aq Nis 2 Ags Ni aq b anode H...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Explain how the following Arduino codes affect the motion of the motor. Which direction is the motor moving? Why the map commands are required? (6%) duty1 = 40; duty2 = 60; dutyl-map...
-
Write the half reactions for the electrolysis of the elements listed in Exercise 3.
-
Write a balanced equation for each of the following reactions or reaction sequences. (a) The reaction catalyzed by PFK-2 (b) The conversion of 2 moles of oxaloacetate to glucose (c) The conversion of...
-
Let z(k) denote the k-year continuously compounded zero-coupon yield for the current term structure. You are given that z(1) = 0.035, z(2) = 0.041, z(3) = 0.045, z(4) = 0.049, z(5) = 0.051, z(6) =...
-
Weighted-average method, assigning costs (continuation of 18-17). For the data in Exercise 18-17, summarize total costs to account for, calculate the cost per equivalent unit for direct materials and...
-
Hugh Leach Corp., a producer of machine tools, wants to move to a larger site. Two alternative locations have been identified: Bonham and McKinney. Bonham would have fixed costs of $800,000 per year...
-
Use It's lemma to express \(d F\) given that \(F(x)=x^{1 / 2}\), where the stochastic process \(\left\{S_{t}, t \geq 0 ight\}\) satisfies the stochastic differential equation \[d...
-
The financial statements of Amazon.com, Inc. are presented in Appendix D. Financial statements for Wal-Mart Stores, Inc. are presented in Appendix E. Instructions (a) Based on the information in the...
-
Solve the system of linear equations, using the Gauss-Jordan elimination method. (If there is no solution, enter NO SOLUTION. If there are infinitely many solutions, express your answer in terms of...
-
A careless laboratory technician wants to prepare 200.0 mL of a 0.025 m HCl(aq) solution but uses a volumetric flask of volume 250.0 mL by mistake. (a) What would the pH of the desired solution have...
-
The concentration of CrO 4 2 in a saturated Tl 2 CrO 4 solution is 6.3 * 10 5 mol L 1 . What is the K sp of Tl 2 CrO 4 ?
-
Molly, whose tax rate is 39.6%, sells an apartment complex for $4.5 million with 10% of the price allocated to land. The apartment complex was purchased in 1993. She has no other sales or exchanges...
-
A particular sine wave has the equation v = 12 sin 0. The rms voltage for this wave is shon formula, sub. V. cale. Refer to the oscilloscope trace in Figure 1. If the VOLTS/DIV control is set to 2...
-
(a) Evaluate the characteristic equation of A = 2 3 -(;;) (b) Let A = 204 98 -26 -10 -134 36 14 348 -90 -36 -232 60 28 1716 -472 Find inverse of A vsing determinant (c) Diagonalize A = 5 - 6 hence...
-
(5.3) This exercise deals with deriving the money multiplier - relating high-powered money (the monetary base) to M1 (ignoring traveler's checks). Money demand for M1, Md, can be decomposed into the...
-
Given a list {Subscript[a, 1],Subscript[a, 2],..,Subscript[a, n]}, its harmonic mean is defined as . Now define three functions hmean1, hmean2 and hmean3, all with the input as a given list and...
-
1. Please look for your assigned unknown in the file titled "Unknown - List and Student Assignments". 2. Then, look for the details of the unknown in the file titled Data for Unknown". 3. Put all of...
-
DigJam Industries specializes in custom printing jobs. The following data pertains to its operations for the most recent year: DigJam writes off any under- or overapplied overhead directly to cost of...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Which of the following functions are eigenfunctions of the operator B if B f (x) = d 2 f (x) / dx 2 : x 2 , cos x, e 3ix ? State the eigenvalue if applicable.
-
Determine in each of the following cases if the function in the first column is an eigenfunction of the operator in the second column. If so, what is the eigenvalue? a. b. c. ei(7x+y) ax? x? + 2y? +...
-
Because d -d cos (nx / d) cos (mx / dx) = 0, m n, the functions cos (n x/d) for n = 1, 2, 3, . . . form an orthogonal set in the interval (d, d). What constant must these functions be multiplied by...
-
Activity Costs Caused by Suppliers ( including failed parts and late delivery ) Activities Costs Repairing Products $ 8 0 0 , 0 0 0 Expediting 2 0 0 , 0 0 0 The Daniels Co Part A 2 0 Part B 2 0...
-
Fees earned for year 1 were $90,000 and for year 2 were $40,000. What was the percent increase or decrease as per the horizontal analysis?
-
You are selected as head of the Environmentally Friendly Methods of Waste Disposal in Commercial Establishments project at the University Teaching Hospital, Lusaka Zambia. Please develop the...

Study smarter with the SolutionInn App


