Write the overall reaction for the mechanism proposed below and identify any reaction intermediates. + CH +
Question:
Write the overall reaction for the mechanism proposed below and identify any reaction intermediates.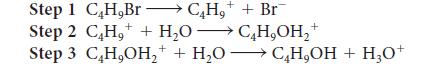
Transcribed Image Text:
+ CH₂ + Br¯ Step 1 C,H,Br + Step 2 C₂H₂ + H₂O →C₂H₂OH₂+ Step 3 C₂H,OH₂++ H₂OC₂H,OH + H₂0+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The mechanism provided shows a threestep reaction sequence To write the overall reaction we need to ...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Write the overall reaction for the mechanism proposed below and identify any reaction intermediates. Step 1 AC + BAB+C Step 2 AC + AB AB + C
-
The following mechanism has been proposed for the gas-phase reaction of H2 with ICl: H2(g) + ICl(g) HI(g) + HCl(g) HI(g) + ICl(g) I2(g) + HCl(g) (a) Write the balanced equation for the overall...
-
The following mechanism has been proposed for the reaction of NO with H2 to form N2O and H2O: NO(g) + NO(g) N2O2(g) N2O2 + H2(g) N2O(g) + H2O(g) (a) Show that the elementary reactions of the...
-
Prepare a statement of profit or loss and other comprehensive income( year ending 31 march 2023) , and a statement of financial position as at 31 march 2023 for the Batliss Plc Bayliss Plc is a...
-
A memory manager can sort the free list according to any criteria it chooses. a. How would the free list be organized for the best-fit policy? b. How would the free list be organized for the...
-
Will Company began operations late in 2013 and adopted the conventional retail inventory method. Because there was no beginning inventory for 2013 and no markdowns during 2013, the ending inventory...
-
Derive the kinematic and dynamic conditions needed in the analysis. Set up the equations to find the constants. Now require that the determinant of the coefficient matrix should be zero to obtain a...
-
Mark Decker has identified four stocks for his portfolio, and he wants to determine the percentage of his total available funds he should invest in each stock. The alternative stocks include an...
-
What is the present value of the following future amount? $311,205 to be received 15 years from now, discounted back to the present at 5 percent, compounded annually.
-
In 1989, a teenager in Ohio was poisoned by breathing vapors from spilled mercury. The mercury level in his urine, which is proportional to its concentration in his body, was found to be 1.54 mg L 1...
-
The iodine clock reaction is a popular chemical demonstration. As part of that demonstration, the I 3 ion is generated in the reaction S 2 O 8 2 (aq) + 3 I (aq) 2 SO 4 2 (aq) + I 3 (aq). In one...
-
The leaf blower draws in air at a rate of 400 ft 3 /min and discharges it at a speed v = 240 mi / hr. If the specific weight of the air being drawn into the blower is 7.53(10 2 ) lb /ft 3 , determine...
-
Think back to one of the largest purchases you've ever made. Discuss which steps in the consumer behavior process were applied and whether cultural, family, or social influences came into play. How...
-
One of the loans included in Sloppy Mortgage Corporations loans at January 1, 2006, was a pay-option ARM loan of \($900,000\) taken out on January 1, 2006, when the value of the house was...
-
The continued acceleration of merger and acquisition (M&A) activity remains strong. As companies find it more challenging to increase revenue growth from within their organizations, big mergers...
-
Which of the following is a signal that a company may be using SPEs to overstate its earnings or understate its debt? (a) When references in the notes to the financial statements regarding...
-
On your own or with a classmate, choose one of the following products and create an advertisement that illustrates how your company creates time, place, and form utility in its delivery of the...
-
Motorolas Finance Department has adapted the concept of delivery cycle time to include the measurement of cycle times for processing customer credit memos, invoices, and orders. Why would such...
-
Cable Corporation is 60% owned by Anna and 40% owned by Jim, who are unrelated. It has noncash assets, which it sells to an unrelated purchaser for $100,000 in cash and $900,000 in installment...
-
Classify each of the following solvents as protic or aprotic: (a) DMF (b) Ethanol (c) DMSO (d) Water (e) Ammonia
-
Consider the following S N 2 reaction: (a) Assign the configuration of the chirality center in the substrate. (b) Assign the configuration of the chirality center in the product. (c) Does this S N 2...
-
Draw the transition state for the reaction between ethyl iodide and sodium acetate
-
1.Irksome Power Co. has learned that its rival utility company has offered to supply a business with electricity for ten years for $50,000 up front and a further $20,000 per year. If Irksome Power...
-
What are Goodwill s competitive advantages having diversity in it's workforce?
-
1. Describe the differences between class A, class B, class AB and class C power amplifiers in the aspects of cycle of operation and efficiency. (10 marks)

Study smarter with the SolutionInn App


