You may need Table 7.2 to answer the following questions. a. Which is the stronger acid, HCl
Question:
You may need Table 7.2 to answer the following questions.
a. Which is the stronger acid, HCl or H2O?
b. Which is the stronger acid, H2O or HNO2?
c. Which is the stronger acid, HCN or HOC6H5
Table 7.2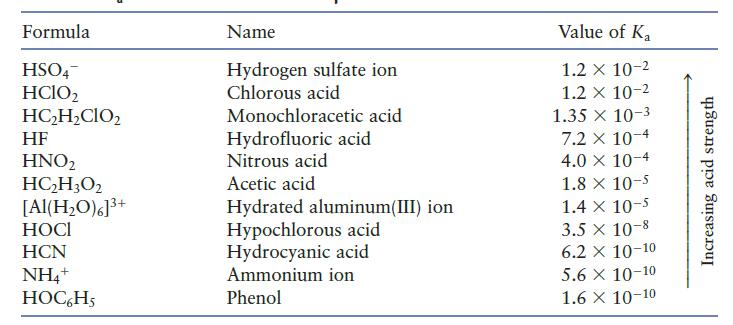
Transcribed Image Text:
Formula HSO4 HClO2 HC₂H₂CIO₂ HF HNO₂ HC₂H3O2 [Al(H₂O)6]³+ HOCI HCN NH4+ HOC6H5 Name Hydrogen sulfate ion Chlorous acid Monochloracetic acid Hydrofluoric acid Nitrous acid Acetic acid Hydrated aluminum(III) ion Hypochlorous acid Hydrocyanic acid Ammonium ion Phenol Value of Ka 1.2 x 10-2 1.2 x 10-2 1.35 x 10-3 7.2 x 10-4 4.0 × 10-4 1.8 x 10-5 1.4 x 10-5 3.5 x 10-8 6.2 X 10-10 5.6 X 10-10 1.6 X 10-10 Increasing acid strength
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
aHCl has higher ka va...View the full answer

Answered By

Godswill Okorie
M.Sc chemistry specialization in organic chemistry, B.ed .I am having industry experience of seven years working with Ranbaxy nd Shimadzu analytical India by working as an application chemistry.I am having good practical experience on chromatography techniques,which later helped me in my teaching.I worked as PGT chemistry teacher with KV and APS.
As a teacher I was able to achieve good results with my students.I used to take 11th and 12th chemistry and science to classes 7th ,8th and ninth. While teaching I used to guide students for various carrier opportunities.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which is the stronger acid in each of the following pairs? Explain your reasoning. (a) Phenol or p-hydroxybenzaldehyde (b) m-Cyanophenol or p-cyanophenol (c) o-Fluorophenol or p-fluorophenol
-
Which is the stronger acid in each of the following pairs?
-
Answer the following questions based on Tables 5P-1 and 5P-2. a. What is the quantity demanded at $10? What is the quantity supplied at $10? b. What is the quantity demanded at $25? What is the...
-
The need to be liked and to stay on good terms with most other people is the need for? a. Affiliation b. Power c. None of the above d. Achievement
-
Suppose that, on a particular day, two persons A and B arrive at a certain store independently of each other. Suppose that A remains in the store for 15 minutes and B remains in the store for 10...
-
Give the names of the following ions. a. Mn 2+ b. Ni 2+ c. Co 2+ d. Fe 3+
-
On February 16, 2005, Joseph and Margaret Buset borrowed $192,000 from Fremont Investment & Loan (the Originator). The loan was secured by a mortgage on a residential condominium. The mortgage named...
-
Corbertt Pharmaceuticals manufactures an over-the-counter allergy medication. The company sells both large commercial containers of 1,000 capsules to health-care facilities and travel packs of 20...
-
Q1. A common base transistor amplifier has an input resistance of 20 Q and output resistance of 100 kn. The collector load is 1 kQ. If a signal of 500 mV is applied between emitter and base, find the...
-
Spherical pellets of 1.0 cm diameter are spray painted with a very thin coat of paint. The paint contains a volatile solvent. The vapor pressure of the solvent at 298 K is 1:17 x 104 Pa, and the...
-
The salt BX, when dissolved in water, produces an acidic solution. Which of the following could be true? (There may be more than one correct answer.) a. The acid HX is a weak acid. b. The acid HX is...
-
Match the following pH values: 1, 2, 5, 6, 6.5, 8, 11, 11 and 13 with the following chemicals (of equal concentration): HBr, NaOH, NaF, NaCN, NH 4 F, CH 3 NH 3 F, HF, HCN, and NH 3 . Answer this...
-
On June 1, 2015, the Crocus Company began construction of a new manufacturing plant. The plant was completed on October 31, 2016. Expenditures on the project were as follows ($ in millions): July 1,...
-
Wellness as the "new sustainability" The notes in this unit introduced the idea that green buildings can have positive effects on workplace productivity and employee health. The article provided to...
-
Discuss the magnitude of COVID-19 government relief money and whether or not the money is going to the correct purpose. What are some of the challenges that public administrators face with handling...
-
On December 31, 2018, Deb's employer gave her a Tag Heuer Limited Edition Tom Brady watch because Deb met her sales quota for the year. Debs employer originally purchased the watch in 2017 for...
-
Given the following program that takes as input the first name of a person and using a switch statement, prints the first and last name together if a match has been found. public class problemOne {...
-
manufacturing costing system uses a? three-part classification of direct? materials, direct manufacturing? labor, and manufacturing overhead costs. Read the requirements Requirement 1. Calculate...
-
Helium gas is compressed from 27C and 3.50 m3/kg to 0.775 m3/kg in a reversible and adiabatic manner. The temperature of helium after compression is (a) 74C (b) 122C (c) 547C (d) 709C (e) 1082C
-
During the year land was revalued and the surplus reported as Revaluation surplus; and an asset costing 80,000, written down to 38,000, was sold for 40,000. Identify the cost of any non-current...
-
A compressed-gas cylinder contains 1.00 10 3 g of argon gas. The pressure inside the cylinder is 2050. psi (pounds per square inch) at a temperature of 18 o C. How much gas remains in the cylinder...
-
Equal moles of sulfur dioxide gas and oxygen gas are mixed in a flexible reaction vessel and then sparked to initiate the formation of gaseous sulfur trioxide. Assuming that the reaction goes to...
-
Silane (SiH 4 ) is the silicon analogue of methane (CH 4 ). It is prepared industrially according to the following equations: Si(s) + 3HCl(g) HSiCl 3 (l) + H 2 (g) 4HSiCl 3 (l) SiH 4 (g) + 3SiCl 4...
-
Misra Inc. forecasts a free cash flow of $50 million in Year 3, ie, at t=3, and it expects FCF to grow at a constant rate of 5.5% thereafter. If the weighted average cost of capital (WACC) is 9.5%...
-
Presentation Outline - Instructions & Rubric make a detailed outline for your PowerPoint presentation. You may write it in point-form if you wish, but ensure that your point form notes are clear and...
-
18. Ms. Smith has two grandchildren, Adam and Evelyn. Adam will be enrolling in college on September 1, 2020, and Evelyn will be enrolling in college on September 1, 2022. Ms. Smith wishes to give...

Study smarter with the SolutionInn App


