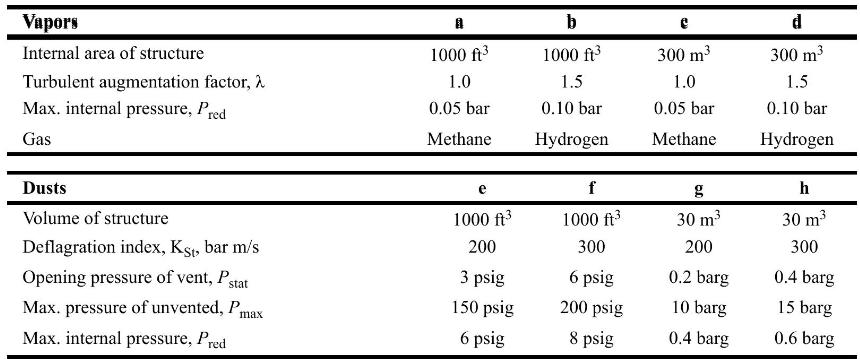
Determine the deflagration vent size for the following structures: Vapors Internal area of structure Turbulent augmentation factor,
Question:
Determine the deflagration vent size for the following structures:
Transcribed Image Text:
Vapors Internal area of structure Turbulent augmentation factor, Max. internal pressure, Pred Gas Dusts Volume of structure Deflagration index, Kst, bar m/s Opening pressure of vent, Pst stat Max. pressure of unvented, P Max. internal pressure, Pred max 8 1000 ft 1.0 0.05 bar Methane e 1000 ft 200 3 psig 150 psig 6 psig b 1000 ft 1.5 0.10 bar Hydrogen f 1000 ft 300 6 psig 200 psig 8 psig 300 m 1.0 0.05 bar Methane 30 m 200 0.2 barg 10 barg 0.4 barg d 300 m 1.5 0.10 bar Hydrogen h 30 m 300 0.4 barg 15 barg 0.6 barg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
We can utilize the formulae supplied by codes like NFPA 68 which relate the deflagration index Kst t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
A 10 -ft-wide by \(10-\mathrm{ft}\)-long by \(10-\mathrm{ft}\)-high shed is used to store tanks of methane. What deflagration vent area is required? Assume a maximum internal overpressure of \(0.1...
-
The cash records and bank statement for the month of July for Ravine Incorporated are shown below. RAVINE INCORPORATED Cash Account Records July 1 , 2 0 2 4 , to July 3 1 , 2 0 2 4 Cash Balance July...
-
Determine whether the following pairs of structures are actually different compounds or simply resonance forms of the same compounds. (a) (b) (c) (d) (e) (f) (g) (i) (j) and O- and and O- CH C H and...
-
using System; using System.Collections.Generic; using System.ComponentModel; using System.Data; using System.Drawing; using System.Linq; using System.Text; using System.Threading.Tasks; using...
-
For the machine element shown, locate the y coordinate of the center of gravity. 135 i
-
In Wageland, all workers sign an annual wage contract each year on January 1. In late January, a new computer operating system is introduced that increases labor productivity dramatically. Explain...
-
The four alternatives described below are being evaluated: The Incremental IRRs are: a. If the alternatives are independent, which one(s) should be selected if \(M A R R=15.5\) percent/year? b. If...
-
Reuse Products, LLC, manufactures plastic beverage bottles. The division that manufactures water bottles for the North American market has two plants that operate 24 hours a day, 365 days a year. The...
-
Recent events have once again shaken and stirred the memory of the nation. Almost immediately after the Civil War, a myth arose painting the conflict with a brush that attempted to blur the lines...
-
A cooling coil contains ethyl alcohol. The heat capacity of the alcohol is \(0.58 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}\), and its density is \(791 \mathrm{~kg} / \mathrm{m}^{3}\). Determine...
-
Determine the relief diameter for the following two-phase flow conditions. Assume in all cases that \(L / D=0.0\). Reaction mass Volume Set pressure Set temperature (dT/dt)s Maximum pressure Maximum...
-
Roll a fair six-sided die. a. What is the probability that the die shows an odd number OR a number greater than 5 on top? b. What is the probability that the die shows an odd number OR a number...
-
We are asked to determine how secure a software lock is. It makes use of the English alphabet of 26 letters. The key to open the lock is FOUR letters long. Upper and lower case are considered a...
-
What is on your mind? What do you want to share? What are you curious about? What do you want the instructor to explain in more detail? What insights do you want to put out there? What crazy,...
-
Assume that a daytime 30-second commercial on Nickelodeon's "Inspector Gadget" costs $9,000 in a particular market. The show has a total target audience of 45,000 children ages ages 6-12. There are...
-
Write a new patient encounter report explaining the concepts below and how you would communicate to the client and perhaps her daughter. Address the following: Explain the importance of autonomy,...
-
GAAP (Generally Accepted Accounting Principles): 1. What is GAAP and why is it important in the field of accounting? 2. What are the core principles of GAAP? 3. How does GAAP impact financial...
-
Best Buy is a specialty retailer of consumer electronics, including personal computers, entertainment software, and appliances. Best Buy operates retail stores in addition to the Best Buy, Media...
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
A 20.0-mL solution of 0.005 00 M Sn 2+ in 1 M HCl was titrated with 0.020 0 M Ce 4+ to give Sn 4+ and Ce 3+ . Calculate the potential (versus S.C.E.) at the following volumes of Ce 4+ : 0.100, 1.00,...
-
Would indigo tetrasulfonate be a suitable redox indicator for the titration of Fe(CN) 6 4- with Tl 3+ in 1 M HCl? The potential at the equivalence point must be between the potentials for each redox...
-
Compute the titration curve for Demonstration 15-1, in which 400.0 mL of 3.75 mM Fe 2+ are titrated with 20.0 mM MnO 4 - at a fixed pH of 0.00 in 1 M H 2 SO 4 . Calculate the potential versus S.C.E....
-
Integrity Lawyers Pty Ltd ('Integrity Lawyers') is a boutique commercial law firm that specialises in complex litigation and class actions in Australia. In light of the recent corporate governance...
-
Convert the decimal number (123.456)10 into the following indicated bases: (a) Binary number (b) Octal number (c) Hexadecimal number (1 Mark) Q2: Convert the decimal number 456 to binary in two ways:...
-
What is the credit rating for Abbott Laboratories bonds? Do all of the bonds have the same credit rating? Why do you think this is ?

Study smarter with the SolutionInn App


