Draw an (F-N) curve using the following data: Incident outcome case Frequency, F; (per year) Estimated number
Question:
Draw an \(F-N\) curve using the following data:
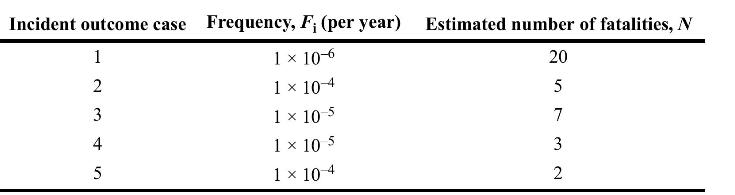
Transcribed Image Text:
Incident outcome case Frequency, F; (per year) Estimated number of fatalities, N 1 x 10-6 20 1 x 10-4 1 x 10 1 10 5 1 10-4 1 2 34 5 -5 57 3 NW 2
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
Ticket to Ride is a popular board game that involves connecting cities in a given railroad network. In this assignment you will prototype some potential approaches for creating an AI player for this...
-
Draw an AON network using the following data and find the probability of completing the critical path of the operatic project in 44 days, the official openingdate. b-420743342423 3 010531222312...
-
a. Approximate f (0.05) using the following data and the Newton forward-difference formula: b. Use the Newton backward-difference formula to approximate f (0.65). c. Use Stirling's formula to...
-
1.3 WANEFMAC7 1.3.007. Find the equation of the given linear function. \[ f(x)= \] WANEFMAC7 1.3.015. Find the slope of the given line, if it is defined. (If an answer is undefined, enter UNDEFINED.)...
-
Locate the centroid of the plane area shown. 10 in. *= ky? 10 in. 15 in.-
-
The following balance sheet and income statement data were taken from the records of L. L. Beeno for the year ended December 31, 2018: Prepare the operating section of the statement of cash flows,...
-
Draw a cash flow diagram of any investment that exhibits both of the following properties: 1. The investment has a 4-year life. 2. The investment has a 10 percent/year internal rate of return.
-
Johnson Chemicals is considering two options for its supplier portfolio. Option 1 uses two local suppliers. Each has a unique- event risk of 5%, and the probability of a super- event that would...
-
Direct Materials Conversion Percent Percent Units Complete Beginning work in process inventory 2,800 100% Complete 25% Units started this period 6,800 Completed and transferred out 7,800 Ending work...
-
The peak overpressure expected as a result of the explosion of a tank in a plant is approximated by the following equation: \[\log P=6.0-1.8 \log r \] where \(P\) is the overpressure in psi and \(r\)...
-
If a regulator has a consequence frequency of \(10^{-1}\) failure \(/ \mathrm{yr}\), what will be the frequency if this regulator is given preventive maintenance once per month?
-
Will the firms in an oligopoly act more like a monopoly or more like competitors? Briefly explain.
-
Summarise what you are going to present in the report and justify your plan for delivering the research project to BIJ. Ensure you also clearly reference a data analytics framework as part of your...
-
Eight years ago Zack& Co. had purchased an equipment fo: $1,200,000. This equipment was being depreciated on a straight line basis over a 12 year period to a 300,000 salvage value. The equipment has...
-
It is a debate that has been raging for weeks. The Pullman City Council voted last night on a motion to approve a controversial sale and improvement project. The vote, which was 4-3, was a close one....
-
Why did the artist address this subject or issue? For what audience and in what situation was it created? Is knowledge of the audience and/or social, historical, and/or cultural situation relevent to...
-
An investor buys 200,000 shares of a private placement that's sold under Regulation D. The investor is not an officer or director of the company. How many shares is the investor permitted to sell...
-
Butte Ironworks Co. reported $7,500,000 for equipment and $6,175,000 for accumulated depreciationequipment on its balance sheet. Does this mean (a) That the replacement cost of the equipment is...
-
What is the role of business risk analysis in the audit planning process?
-
We will abbreviate malonic acid, CH 2 (CO 2 H) 2 , as H2M. Find the pH and concentrations of H 2 M, HM + , and M 2+ in (a) 0.100 M H 2 M; (b) 0.100 M NaHM; (c) 0.100 M Na 2 M.
-
Starting with the fully protonated species, write the stepwise acid dissociation reactions of the amino acids glutamic acid and tyrosine. Be sure to remove the protons in the correct order. Which...
-
Draw the structure of the predominant form of pyridoxal-5-phosphate at pH 7.00.
-
Robbins Corp. frequently invests excess funds in the Mexican money market. One year ago, Robbins invested in a one - year Mexican money market security that provided a yield of 2 5 percent. At the...
-
Compare and Contrast business ethics and social responsibility. 2- Describe Kohlberg's three stages of personal ethical development. 3- Detail each of the four rights of consumers as put forward by...
-
Suppose you take a 3 / 1 interest - only, 3 0 - year ARM for $ 1 8 0 , 0 0 0 . 0 0 with an interest - only contract rate of 3 % and a following rate of 7 . 7 5 % in year four. Assume you pay 1 . 5...

Study smarter with the SolutionInn App


