A step tracer input was used on a real reactor with the following results: a. What is
Question:
A step tracer input was used on a real reactor with the following results: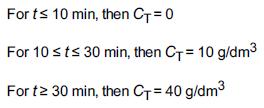
a. What is the mean residence time tm?
b. What is the variance σ2?
Transcribed Image Text:
For t≤ 10 min, then C₁ = 0 For 10 st≤ 30 min, then C+= 10 g/dm³ For t≥ 30 min, then C+= 40 g/dm³
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a The cumulative distributio...View the full answer

Answered By

Muhammad Mahtab
everyone looks that their work be perfect. I have more than a five year experience as a lecture in reputable institution, national and international. I provide perfect solution in marketing, case study, finance problems, blog writing, article writing, business plans, strategic management, human resource, operation management, power point presentation and lot of clients need. Here is right mentor who help clients in their multi-disciplinary needs.
5.00+
3+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Doing the computations by hand, find the determinant of the matrix below. 6 2
-
Answer Exercise 1 for the case where α is replaced by 4i 3 2
-
Use the RTD data in Examples 16-1 and 16-2 to predict X PFR , X CSTR , X LFR , X T-I-S , X seg and X mm for the following elementary gas-phase reactions a. A B k = 0.1 min 1 b. A 2B k = 0.1 min 1...
-
Solve the right triangles with the given parts or state that there is not enough information to solve. Round off results according to Table 4.1. Refer to Fig. 4.37. B = 32.1, c = 238 Data from Table...
-
In July 1988, Chester Crow executed a promissory note payable to the order of THE FIRST NATIONAL BANK OF SHREVEPORT or BEARER in the amount of $21,578.42 at an interest rate of 3 percent per year...
-
The effective rate constant for a gaseous reaction that has a Lindemann-Hinshelwood mechanism is 1.7 X 10-3 s-I at 1.09 kPa and 2.2 X 10-4 S-1 at 25 Pa. Calculate the rate constant for the activation...
-
Consider two alternatives, each of which will accomplish the same EPA-mandated pollution control. Using an incremental IRR analysis, determine the preferred alternative assuming MARR is 20...
-
Ramiro & Sons buys T-shirts in bulk, applies its own trendsetting silk-screen designs, and then sells the T-shirts to a number of retailers. Ramiro wants to be known for its trendsetting designs, and...
-
Sales for a new car is expected to grow according to the equation: S = 150000(1-e -0.06t ), where t = months i) Calculate the number of cars sold after one year. ii) Calculate the number of cars sold...
-
Big Guy Inc. purchased 80% of the outstanding voting shares of Humble Corp. for $360,000 on July 1, 2017. On that date, Humble Corp. had common shares and retained earnings worth $180,000 and...
-
The following E(t) curves were obtained from a tracer test on two tubular reactors in which dispersion is believed to occur. (a) RTD Reactor A; (b) RTD Reactor B The graphs for RTD reactors A and B...
-
Consider the E(t) curve below. A graph is shown, with t (in minutes) on horizontal axis and E of t (minutes inverse) on vertical axis. A hemi (half) circular curve starts at the origin and ends at 2...
-
Major corporations with overseas subcontractors (such as IKEA in Bangladesh, Unilever in India, and Nike in China) have been criticized, often with substantial negative publicity, when children as...
-
The Lopes Activity Tracker will be used to document and follow the status of your practice immersion hours throughout this and every subsequent course. Please review the Practice Immersion Hours...
-
. Debit A. The cost of operating a business; a decrease in stockholders' equity 2. Expense B. Assets - Liabilities 3. Net income C. Grouping of accounts 4. Ledger D. Copying data from the journal to...
-
Preston has been working for Thompson Industries for just over a year and has been part of Thompson's Deferred Profit Sharing Plan (DPSP) program from his start date. The program has a 2-year vesting...
-
The company has three preparation In a separate worksheet, reformulate the problem (you can just change the model you created in excel, in part b) to determine if there is an alternative optimal...
-
Use the function c to create a vector with the average temperatures in January for the cities A, B, C, D, E, and F, which are 35, 88, 42, 84, 81, and 30 degrees Fahrenheit, respectively. Call the...
-
For the past several years, Jeff Horton has operated a part-time consulting business from his home. As of April 1, 2014, Jeff decided to move to rented quarters and to operate the business, which was...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
Why do we say that the equilibrium constant for the reaction H 2 O H + + OH - (or any other reaction) is dimensionless?
-
Write the expression for the equilibrium constant for each of the following reactions. Write the pressure of a gaseous molecule, X, as P X . a. b. 3Ag*(aq) + PO (aq) = Ag3PO4(s)
-
(a) A favorable entropy change occurs when S is positive. Does the order of the system increase or decrease when S is positive? (b) A favorable enthalpy change occurs when H is negative. Does the...
-
Rewrite as an exponential equation. lny = 6 Olog In X =
-
If y = sin (u), u = v-, and v = ln (x), then the value of dy dz at e is 49 01 Oo 22
-
Solve. 6x - 17x + 5 = 0

Study smarter with the SolutionInn App


