The aqueous decomposition of A is studied in an experimental mixed flow reactor. The results in Table
Question:
The aqueous decomposition of A is studied in an experimental mixed flow reactor. The results in Table P5.25 are obtained in steady-state runs. To obtain 75% conversion of reactant in a feed, CA0 = 0.8 mol/liter, what holding time is needed in a plug flow reactor?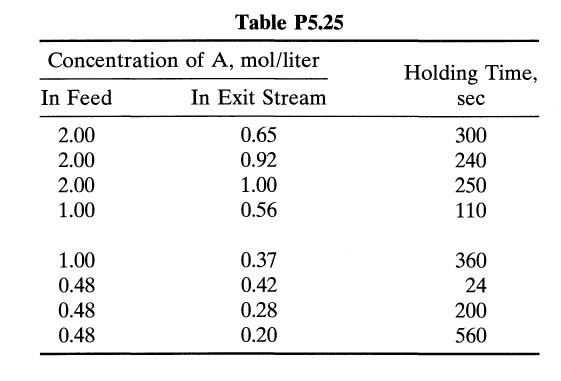
Transcribed Image Text:
Concentration In Feed 2.00 2.00 2.00 1.00 1.00 0.48 0.48 0.48 Table P5.25 of A, mol/liter In Exit Stream 0.65 0.92 1.00 0.56 0.37 0.42 0.28 0.20 Holding Time, sec 300 240 250 110 360 24 200 560
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine the required holding time in a plug flow reactor to achieve 75 conversion of reactant A ...View the full answer

Answered By

Prachi Goyal
No experience of tutoring and teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Repeat the previous problem but for a mixed flow reactor. Previous Problem The aqueous decomposition of A is studied in an experimental mixed flow reactor. The results in Table P5.25 are obtained in...
-
The aqueous decomposition of A produces R as follows: The following results are obtained in a series of steady state runs, all having no R in the feed stream. From this kinetic information, find the...
-
The kinetics of the aqueous-phase decomposition of A is investigated in two mixed flow reactors in series, the second having twice the volume of the first reactor. At steady state with a feed...
-
QUESTION 7 We discussed the selection sort sorting algorithm. What is the worst case time complexity of selection sort when sorting a list of n elements? o(lg n) O(n) O(n lg n) O(n) O(log n) O(1)
-
The manager for Tyler Bank and Trust has the following assets and liabilities to manage: If the manager wants a duration gap of 3.00, what level of saving accounts should the bank raise? Assume that...
-
Four point masses of 3.0 kg each are arranged in a square on massless rods. The length of a side of the square is 0.50 m. What is the rotational inertia for rotation about an axis (a) Passing through...
-
For each of the following situations, calculate the population standard error of the mean \(\sigma \mathrm{X}^{-}\). a. \(\sigma=8 ; N=16\) b. \(\sigma=12 ; N=64\) c. \(\sigma=2 ; N=25\) d....
-
The National Center for Education Statistics reported that 47% of college students work to pay for tuition and living expenses. Assume that a sample of 450 college students was used in the study. a....
-
Turner Company owns 15% of the outstanding stock of ICA Company. During the current year, ICA paid a $4.70 million cash dividend on its common shares. Ignore income taxes. What effect did this...
-
The data in Table P5.28 have been obtained on the decomposition of gaseous reactant A in a constant volume batch reactor at 100C. The stoichiometry of the reaction is 2A R + S. What size plug flow...
-
A high molecular weight hydrocarbon gas A is fed continuously to a heated high temperature mixed flow reactor where it thermally cracks (homogeneous gas reaction) into lower molecular weight...
-
You have been asked to do an in-depth analysis of a company's financial statements but you haven't been told what the purpose of the analysis is-that is, what decision needs to be made. Is it...
-
How can you highlight compnents of a formula when working inside a cell? Group of answer choices Ctrl Shift $ Ctrl Shift Enter Ctrl Shift ( ( Arrow ) )
-
Question 4 The following are the cash-book and bank statement of P. Thomas for the month of November 2022. Bank Columns Only Cash Book 2022 $ November 1 balance b/d 2,379 November November 7 B Green...
-
(b) Answer BOTH parts (i) and (ii). (i) Suggest a structure for A and provide a mechanism for its formation. i). NaOEt/EtOH ii). CH3-1 A 6H8O2 NaOD/DO (ii) Suggest a structure for B and account for...
-
This project requires the use of a Windows, Linux, or Mac computer that has Internet access. Using methods you learned in this chapter, find the answers to the following questions: 1 1 How is an IP...
-
Question: Which initiative exploits the software-defined networking implementation to enhance network security by tightly controlling the environment, often using virtualization and terraforming?...
-
Jensens Juice Bar is considering purchasing a new blender. Indicate which of the following statements a relevant consideration in the new-blender decision. a. Last year, Jensens spent $500 on a new...
-
Does log 81 (2401) = log 3 (7)? Verify the claim algebraically.
-
The irreversible gas-phase dimerization 2A A2 is carried out at 8.2 atm in a stirred contained-solids reactor to which only pure A is fed. There are 40 g of catalyst in each of the four spinning...
-
The second-order decomposition reaction A B + 2C is carried out in a tubular reactor packed with catalyst pellets 0.4 cm in diameter. The reaction is internal-diffusion-limited. Pure A enters the...
-
A first-order reaction is taking place inside a porous catalyst. Assume dilute concentrations and neglect any variations in the axial (x) direction. a. Derive an equation for both the internal and...
-
If the absolute pressure of the air in an engine cylinder is 100 kPa (14.5 psi) at the beginning of the compression stroke, what is the air pressure at the end of the stroke if the air is compressed...
-
Problem 3 The linkage shown lies in the horizontal plane. End A of the bar follows the horizontal guide, and a wheel at B is aimed at an angle b that is a specified function of time. The wheel rolls...
-
1. Identify four (4) important items of personal safety when carrying diagnostic procedures on vehicles? 1 2 3 4 2. What precautions should be taken before disconnecting or removing a component of an...

Study smarter with the SolutionInn App


