A nitrogen gas laser pulse with a wavelength of 337 nm contains 3.83 mJ of energy. How
Question:
A nitrogen gas laser pulse with a wavelength of 337 nm contains 3.83 mJ of energy. How many photons does it contain?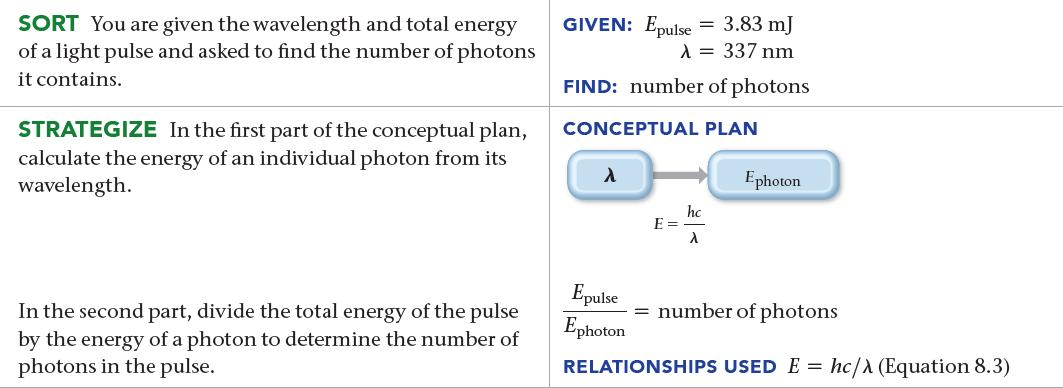
Transcribed Image Text:
SORT You are given the wavelength and total energy of a light pulse and asked to find the number of photons it contains. STRATEGIZE In the first part of the conceptual plan, calculate the energy of an individual photon from its wavelength. In the second part, divide the total energy of the pulse by the energy of a photon to determine the number of photons in the pulse. GIVEN: Epulse = 3.83 mJ λ = 337 nm FIND: number of photons CONCEPTUAL PLAN λ E = hc λ Ephoton Epulse Ephoton = number of photons RELATIONSHIPS USED E= hc/> (Equation 8.3)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
337 nm X Ephoton hc 10 m 1 nm 337 x 107 m 6626 X 1034Js 3...View the full answer

Answered By

Mishark muli
Having any assignments and any other research related work? worry less for I am ready to help you with any task. I am quality oriented and dedicated always to produce good and presentable work for the client once he/she entrusts me with their work. i guarantee also non plagiarized work and well researched work to give you straight As in all your units.Feel free to consult me for any help and you will never regret
4.70+
11+ Reviews
37+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A 1-second pulse of a red laser pointer with a wavelength of 635 nm contains 5.0 mJ of energy. How many photons does it contain?
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
A laser used to weld detached retinas puts out 28-ms-long pulses of 640-nm light which average 0.68-W output during a pulse. How much energy can be deposited per pulse and how many photons does each...
-
(1) Choose all of the following statements that are correct about the time evolution of a general wave function: (I) The time evolution of a general wave function is governed by the Hamiltonian...
-
In its 2006 annual report, McDonalds Corporation reports beginning total assets of $30.0 billion; ending total assets of $29.0 billion; net sales of $21.6 billion, and net income of $3.5 billion. (a)...
-
What is materiality? Why is it an important concept for auditors to consider in the conduct of their audits?
-
Using Statistical Sampling to Achieve Audit Program Steps Robin Hamilton, a first-year staff accountant, is reviewing the following selected audit procedures scheduled for use during the...
-
Herbert, Inc., acquired all of Rambis Companys outstanding stock on January 1, 2014, for $574,000 in cash. Annual excess amortization of $12,000 results from this transaction. On the date of the...
-
You have been watching the "Zero Gravity" ride at the Del Mar Fair (unfortunately not this year, though), where an individual is pinned against the back wall with enough force to hold them up as the...
-
Your company is considering deploying a chatbot, a virtual customer assistant, to initially handle customer service calls and, only when necessary, route callers to human operators for additional...
-
Which wavelength of light has the highest frequency? a) 10 nm b) 10 mm c) 1 nm d) 1 mm
-
Which statement best describes the differences between a bright green laser and a dim red laser? (a) The two lasers emit light of the same frequency, and the light from the green laser has a greater...
-
Alice is single and self-employed in 2020. Her net business profit on her Schedule C for the year is $150,000. What is her self-employment tax liability and additional Medicare tax liability for 2020?
-
Mountain tops applies overhead on the basis of direct labor hours and reports the following information: Budget Actual Overhead $ 4 5 0 , 0 0 0 $ 4 5 2 , 0 0 0 Direct Labor Hours 7 5 , 0 0 0 7 7 , 0...
-
Margaret, a married taxpayer filing a joint return, engaged in two business activities this year. Business A earned $ 4 0 0 , 0 0 0 of profit. Business B incurred a loss of $ ( 9 9 5 , 0 0 0 ) ....
-
1. For the following generating functions list all formal products in which the exponent sums to 4. (a) f(x) = (1+r + x 4 ) 2 (1 + x + x) 2 (b) g(x)=(1+x + x + x + x +x + ...) 3. 2. Build a...
-
Grixdale Tax Services prepares taxes for individuals. Grixdale offers a simplified pricing model with two alternatives for taxpayers: Standard Deduction ( Standard ) or Itemized Deductions ( Itemized...
-
what ways can an individual demonstrate empathetic resonance with diverse perspectives and experiences ?
-
a. Explain the meaning of risk control. b. Explain the following risk-control techniques. 1. Avoidance 2. Loss prevention 3. Loss reduction
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
Show that two of the set of four equivalent orbitals appropriate for sp 3 hybridization, And are orthogonal. W. =-62, + 2p, + 2, + 2p,) (: - O2p. 2p, + 2p,)
-
Show that the water hybrid bonding orbitals given by a = 0.55 2Pz + 0.71 2 px -0.45 2 ps b = 0.55 2pz - 0.71 2px - 0.45 2s are orthogonal.
-
Predict which of the bent molecules, BH 2 or NH 2 , should have the larger bond angle on the basis of the Walsh correlation diagram in Figure 24.11. Explain your answer. Figure 24.11 16, 1Tu + 2a,...
-
A competitor, Beta Electronics, sets up an identical factory (same fixed and variable costs) producing y thousand chips per month. (a) (1 mark) Find P(x, y) and Q(x, y), the profit functions for the...
-
If the situation could have been prevented before it got worse, how should the managers have done differently? Discuss actions by Abigail's former manager, HR specialist, and Mike. State what they...
-
An automobile tire is inflated with air originally at 10.0C and normal atmospheric pressure. During the process, the air is compressed to 27.0% of its original volume and the temperature is increased...

Study smarter with the SolutionInn App


