A solution contains equal concentrations of Ba 2+ , Pb 2+ , and Ca 2+ ions. When
Question:
A solution contains equal concentrations of Ba2+, Pb2+, and Ca2+ ions. When postassium sulfate is added to this solution, which cation precipitates first?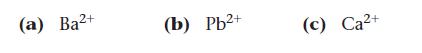
Transcribed Image Text:
(a) Ba²+ (b) Pb²+ (c) Ca²+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Ba has the smallest solubil...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Generate a random variable from the p.d.f f(x)=2x:0x1 and f(x)=0:x0x1 using acceptance rejection method. Take g(x)=1,0x1 and c=2
-
A saturated solution of copper(I) sulfide, Cu 2 S, contains 1.91 10 12 g of Cu 2 S dissolved in 1 dm 3 of water. (A r values: Cu = 63.5, S = 32.1) a. Write an equilibrium expression for the...
-
Silver sulfate dissolves in water according to the reaction: A 1.5-L solution contains 6.55 g of dissolved silver sulfate. If additional solid silver sulfate is added to the solution, will it...
-
In SSL and TLS, why is there a separate Change Cipher Spec Protocol rather than including a change_cipher_spec message in the Handshake Protocol?
-
On January 2, 2013, Benson Hospital purchased a $100,000 special radiology scanner from Picard Inc. The scanner had a useful life of 4 years and was estimated to have no disposal value at the end of...
-
More Complex Financial Statements Industrial Relations Ltd. offers mediation services for unionized companies. When there is a contract dispute between a union and company management, often the two...
-
Oncology Tech is a manufacturer of cancer therapy devices called compensators. CMS was a company that sold cancer treatment planning software for use in purchasing cancer treatment devices. In 2006,...
-
At the beginning of the year, Plummer's Sports Center bought three used fitness machines from Advantage, Inc. The machines immediately were overhauled, installed, and started operating. The machines...
-
asap please 1. Determine a maintenance priority by calculating Preventive Maintenance Factors (PMFs) for the three pieces of equipment listed in the table below. Clearly state which piece of...
-
Determine whether each compound is more soluble in an acidic solution than it is in a neutral solution. (a) BaF 2 (b) AgI (c) Ca(OH) 2
-
Three 15.0-mL acid samples0.10 M HA, 0.10 M HB, and 0.10 M H 2 Care all titrated with 0.100 M NaOH. If HA is a weak acid, HB is a strong acid, and H 2 C is a diprotic acid, which statement is true of...
-
A payday loan is structured to obscure the true interest rate you are paying. For example, in Washington, you pay a $33 fee for a two-week $200 payday loan (when you repay the loan, you pay $233)....
-
Use the information given in question 38 to test the hypothesis that the population mean accounts receivable is greater than $200 at the .05 level of significance. Question 38 An auditor is...
-
Suppose a random sample of 25 people at a local weight-loss center is taken and the mean weight loss is found to be 12 lb. From past history, the standard deviation is known to be 3 lb. Test the...
-
An investment advisor is interested in determining whether a retirement community represents a potential clientele base. Of the 2,000 residents, he randomly samples 100 individuals and finds their...
-
Write pros and cons of Heap-Sort, Merge-Sort and Quick-Sort.
-
Use the information given in question 53 and test, at the 5 % level of significance, the alternative hypothesis that the mean weight loss is at least 4 lb. Question 53 A diet center claims that...
-
Listed below are three different products in various stages of the product life cycle. What marketing strategies would you suggest to these companies? (a) Canon digital camerasgrowth stage, (b)...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
Sketch Bode magnitude and phase plots for 1.6 s = jo H(s) = s(s + s + 16)
-
Construct the Bode magnitude and phase plots for 2(s + 1) H(s) = s = ja (s + 2)(s + 10)*
-
Draw the Bode plots for 250(jo + 1) jo(-a + 10 ja + 25) ) -
-
Each of the three independent situations below describes a finance lease in which annual lease payments are payable at the beginning of each year. The lessee is aware of the lessor's implicit rate of...
-
The following selected transactions were taken from the books of Ripley Company for Year 1: 1. On February 1, Year 1, borrowed $70,000 cash from the local bank. The note had a 6 percent interest rate...
-
14. Verrett Corporation is a manufacturer that uses job-order costing. The company has supplied the following data for the just completed year: Raw materials purchased on account Raw materials (all...

Study smarter with the SolutionInn App


