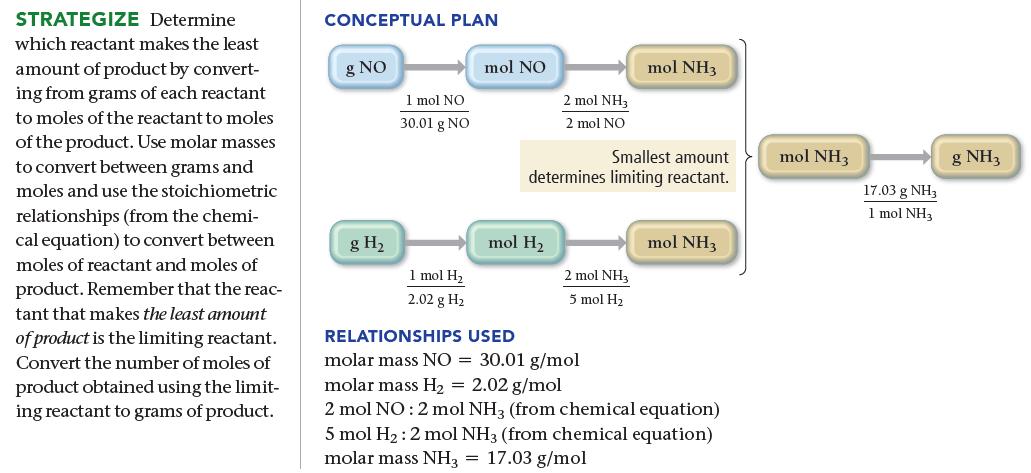
Ammonia, NH 3 , can be synthesized by the reaction: Starting with 86.3 g NO and 25.6
Question:
Ammonia, NH3, can be synthesized by the reaction:![]()
Starting with 86.3 g NO and 25.6 g H2, find the theoretical yield of ammonia in grams.
Transcribed Image Text:
2 NO(g) + 5 H₂(g) 2 NH3(g) + 2 H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
863 g NO X Limiting reactant 1 mol NO 2 mol NH3 3001 g NO 2 melNO 256 ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Ammonia, NH3, can be synthesized by the reaction: 2 NO(g) + 5 H2(g) -------> 2 NH3(g) + 2 H2O(g) Starting with 96.4 g NO and 35.8 g H2, find the theoretical yield of ammonia in grams.
-
Nitrogen dioxide NO 2 can be synthesized by the reaction: N 2 (g) + 2 O 2 (g) 2 NO 2 (g) Starting with 46.3 g N 2 and 92.0 g O 2 , find the theoretical yield of NO 2 in grams.
-
The following compounds can be synthesized by aldol condensations, followed by further reactions. (In each case, work backward from the target molecule to an aldol product, and show what compounds...
-
A company has the following data: net sales, $405,000; cost of goods sold, $220,000; selling expenses, $90,000; general and administrative expenses, $60,000; interest expense, $4,000; and interest...
-
The financial statements of Tootsie Roll Industries are presented in Appendix A. Instructions Answer the following questions. (a) What was the amount of net cash provided by operating activities for...
-
Using the method of superposition, determine the magnitude of \(\mathbf{M}_{0}\) in terms of the distributed load \(w\) and dimension \(a\) so that the deflection at the center of the beam is zero....
-
1. How many documents does ThyssenKrupp have to process to build one elevator? 2. How did using M-Files benefit ThyssenKrupp? 3. What consequence did ThyssenKrupp face because of project delays? 4....
-
The Dauve Company reported the following condensed income statement for 2007: During 2007, the following changes occurred in the company's current assets and current liabilities: Increase (Decrease)...
-
Table 1 describes the bid history for some past auction on eBay. The starting price of the auction was $0.99, and the final price (paid by the winner, bidder D) was $250.00. 1. Find the missing...
-
Angie March owns a catering company that stages banquets and parties for both individuals and companies. The business is seasonal, with heavy demand during the summer months and year-end holidays and...
-
Nitrogen and hydrogen gas react to form ammonia according to the reaction: A flask contains a mixture of reactants represented by the image shown at the left. Which of the following images best...
-
Sodium and chlorine react to form sodium chloride: What is the theoretical yield of sodium chloride for the reaction of 55.0 g Na with 67.2 g Cl 2 ? a) 1.40 * 10 2 g NaCl b) 111 g NaCl c) 55.4 g NaCl...
-
A firm often obtains services from subsidiaries in which the firm's key officers may hold minority ownership. What incentive conflicts do such arrangements pose?
-
On February 1, 2022, Messi Inc. purchased a patent from a research firm by issuing 20,000 shares of its $1 par value common stock. On the date of purchase, the stock was trading on a public exchange...
-
Camille Sikorski was divorced in 2018. She currently provides a home for her 15-year-old daughter Kaly. Kaly lived in Camille's home for the entire year, and Camille paid for all the costs of...
-
Calculate (426) +204 5-6
-
6.) Farrah Singh will be reviewing the federal fund rate of each bank as well. Why is the federal fund rate important when determining the medical facility's banking needs
-
7. If your original loan amount for a 30-year fixed rate Interest Only (IO) mortgage loan is $300,000?, and the annual interest rate is 10%, what is the monthly payment
-
Define each of the following: a. Products liability b. Completed operations liability c. Contractual liability d. Contingent liability
-
Citing a scientific article, explain in your own words, how DNA fingerprinting has been used in forensic science to solve crimes and why it may not always be accurate or effective.
-
You have collected a tissue specimen that you would like to preserve by freeze drying. To ensure the integrity of the specimen, the temperature should not exceed 5.00C. The vapor pressure of ice at...
-
The phase diagram of NH 3 can be characterized by the following information. The normal melting and boiling temperatures are 195.2 and 239.82 K, respectively; the triple point pressure and...
-
Use the vapor pressures of ice given here to calculate the enthalpy of sublimation using a graphical method or a least squares fitting routine. T (K) P (Torr) 200. 0.1676 210. 0.7233 2.732 220. 230....
-
(b) The typical de Broglie wavelength, , is given as 2 = h 3mkBT where m is the mass of a particle, kB is the Boltzmann's constant and T is the temperature in Kelvin. If the lattice spacing in a...
-
Aging Infrastructure - The City of Toronto has some of the oldest buildings in Ontario, which presents a challenge for firefighters in terms of accessibility and safety. Provincial Issue: Funding -...
-
Part A Two point charges are placed on the z axis as follows: Charge q = +4.05 nC is located at x=0.200 m and charge q2=-5.40 nC is at x = +0.400 m. What is the magnitude of the net force exerted by...

Study smarter with the SolutionInn App


