An aspirin tablet contains 325 mg of acetylsalicylic acid (C 9 H 8 O 4 ). How
Question:
An aspirin tablet contains 325 mg of acetylsalicylic acid (C9H8O4). How many acetylsalicylic acid molecules does it contain?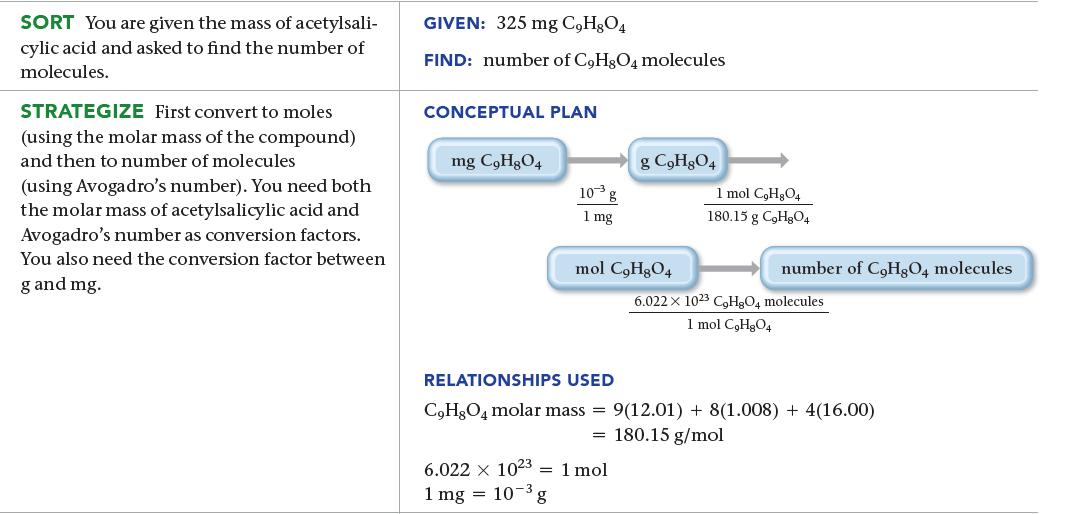
Transcribed Image Text:
SORT You are given the mass of acetylsali- cylic acid and asked to find the number of molecules. STRATEGIZE First convert to moles (using the molar mass of the compound) and then to number of molecules (using Avogadro's number). You need both the molar mass of acetylsalicylic acid and Avogadro's number as conversion factors. You also need the conversion factor between g and mg. GIVEN: 325 mg C,H,O4 FIND: number of C9H8O4 molecules CONCEPTUAL PLAN mg C₂H₂O4 10³ g 1 mg g C₂H8O4 mol C₂H₂O4 6.022 x 1023 1 mol 1 mg = 10-3, ³g 1 mol C₂H₂O4 180.15 g C₂H8O4 number of CoHgQ4 molecules 6.022 x 1023 C₂H8O4 molecules 1 mol C₂H₂O4 RELATIONSHIPS USED C₂H8O4 molar mass= 9(12.01) + 8(1.008) + 4(16.00) = 180.15 g/mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
325 mg CH8O4 X 10 g 1 m...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A typical aspirin tablet contains 325 mg acetylsalicylic acid (HC 9 H 7 O 4 ). Calculate the pH of a solution that is prepared by dissolving two aspirin tablets in enough water to make one cup (237...
-
Given f(x) is a polynomial function with leading term of -24x^(11), a. How many zeros will f(x) have? b. How many x-intercept s can f(x) have? c. How many turning points will f(x) have?
-
Given a rigid tank with V=800 L filled with m = 2 kg of HO at P = 0.2 MPa. Write your answers in the table below. Only answers in the table are counted. Specific volume (m/kg) - R Phase description:...
-
What is marginal cost Explain with an example.
-
In the previous problem, suppose your required return on the project is 20 percent and your pretax cost savings are $340,000 per year. Will you accept the project? What if the pretax cost savings are...
-
The parabolic spandrel shown is revolved about the x axis to form a homogeneous solid of revolution of mass m, using direct integration, express the moment of inertia of the solid with respect to the...
-
Harold Fail owned a plot of land in Georgia known as Ebenezer Landing, where he operated a public boat ramp and parking lot. In 1999, Fail and Steve Bodiford entered into an oral agreement...
-
Data for Nosker Company are presented in P17-7A. Further analysis reveals the following. In P17-7A, Presented below are the financial statements of Nosker Company. NOSKER COMPANY Income Statement For...
-
28. Below is the model of a three-dimensional structure. Draw the front view of this model. Each cube is exactly inch in length, width, and height. Use a scale of 2:1. (2 marks) Back Top Front Left...
-
1. Array Subsets Given an integer array, divide the array into 2 subsets A and B while respecting the following conditions: The intersection of A and B is null. The union A and B is equal to the...
-
What is the formula mass for a compound? Why is it useful?
-
A compound has the empirical formula CH 2 O and a formula mass of 120.10 amu. What is the molecular formula of the compound? a) CH 2 O b) C 2 H 4 O 2 c) C 3 H 6 O 3 d) C 4 H 8 O 4
-
What have you learned from the research on work centrality and the relative importance of work dimensions to people around the world?
-
A force of 14.0 N directed down a ramp inclined at 20 pushes on a 19.5 kg box; the box is at the point where it is just about to move. Determine the coefficient of static friction between the box and...
-
calculate the current yield for the following bonds: Bond: Coupon YTM (annualized) Maturity A B 7.00% 4.50% 5.62% 5.00% 5 years 5 years Frequency: (Semiannual Payments) (Non-callable) 2 2...
-
A hospital wants to purchase a diagnostic system for $500,000 and depreciate it on a straight-line basis over a five year period for tax purposes. The investment would result in revenues of $225,000...
-
Briefly describe the difference between the common law and civil law traditions. (2 marks) What are the four essential elements of a valid contract in both the common law and civil law...
-
Describe characteristics of important people in an organization's external environment (i.e. traditions, lifestyles, values, attitudes, beliefs, opinions, tastes, political views, and behavioral...
-
Rehab Physical Therapy Inc. is planning its cash payments for operations for the second quarter (MarchMay), 2013. The Accrued Expenses Payable balance on March 1 is $36,000. The budgeted expenses for...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
For 1.25 mol of an ideal gas, P external = P =350. 10 3 Pa.The temperature is changed from 135C to 21.2C, and C V ,m = 3/2R. Calculate q, w, U, and H.
-
Suppose an adult is encased in a thermally insulating barrier so that all the heat evolved by metabolism of foodstuffs is retained by the body. What is her temperature increase after 2.5 hours?...
-
Draw bond-line structures for all constitutional isomers of C 5 H 12 ?
-
2 1. Let I = S 1 + exc - - ex -dx. 1+ ex 0
-
The accompanying tree diagram represents an experiment consisting of two trials. Use the diagram to find the given probability. P(Cn E) E 0.8 A 0.2 0.2 EC 0.5 E 0.5 B 0.5 EE EC 0.3 0.3 C 0.7 E
-
2 The Fourier series of the square signal shown below is: 2 f(t) = sin(wo) + [3Pts] 2 2 2 sin(3w0) + sin(5w0) + sin(7wo) + -sin(9wo) T 3 5 7 9 2 + sin(11w0) + ... 11 Plot the first five terms of the...

Study smarter with the SolutionInn App


