An electrochemical cell is based on these two half-reactions: Calculate the cell potential at 25 C. Ox:
Question:
An electrochemical cell is based on these two half-reactions: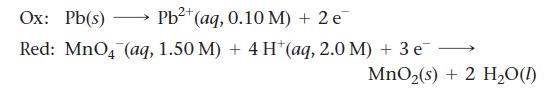
Calculate the cell potential at 25 °C.
Transcribed Image Text:
Ox: Pb(s) Pb²+ (aq, 0.10 M) + 2 e Red: MnO4 (aq, 1.50 M) + 4H* (aq, 2.0 M) + 3 e MnO₂ (s) + 2 H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

BRIAN MUSINGA
I possess a Bachelors of Commerce degree(Marketing option) and am currently undertaking an MBA in marketing. I believe that I possess the required knowledge and skills to tutor in the subject named. I have also written numerous research academic papers much to the satisfaction of clients and my professors.
5.00+
2+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An electrochemical cell is constructed such that on one side a pure nickel electrode is in contact with a solution containing Ni2+ ions at a concentration of 3 10-3 M. The other cell half consists...
-
The reaction taking place in an electrochemical cell under standard conditions is Fe 2+ (aq) + Ag + (aq) Fe 3+ (aq) + Ag(s) a. Write two half-equations for this reaction. For each, state whether...
-
An electrochemical cell is based on the following two half-reactions: Ox: Pb(s)Pb2+(aq,Pb(s)Pb2+(aq, 0.15 MM )+2e)+2e Red: MnO4(aq,MnO4(aq, 1.80 MM )+4H+(aq,)+4H+(aq, 1.9 MM )+3e)+3e MnO2(s)+2H2O(l)...
-
Stock W, X and Y have expected returns of 9.0%, 16.1% and 11.3% respectively. Based on this and the attached information, what is the expected return of your portfolio? Stock W X Y Number of Shares...
-
Last year, Biomed Laboratories, inc., researched and perfected a cure for the common cold. Called Cold-Gone, the product sells for $28.00 per package, each of which contains five tablets. Standard...
-
A recent survey of financial executives found that they favor stock repurchases over dividends. How does that finding seem to contradict the idea that firms use distribution decisions to signal...
-
In spring 1989, Michael Jordan and the Chicago Bulls were in Indianapolis, Indiana, to play against the Indiana Pacers. At the same time, Karla Knafel was singing with a band at a hotel in...
-
The bank portion of the bank reconciliation for Langer Company at November 30, 2017, was as follows The adjusted cash balance per bank agreed with the cash balance per books at November 30. The...
-
A student performs a double-slit experiment using two slits spaced 0.20 mm apart and located 1.50 m from the screen. The monochromatic light source creates an interference pattern in which the...
-
Go to the following World Bank webpage: http://www.app.collinsindicate.com/worldbankatlas-global/en-us In the search box in the upper right corner of the page, enter the following: GDP per capita,...
-
A voltaic cell employs the following redox reaction: Calculate the cell potential at 25 C under each set of conditions. Sn+ (aq) + Mn(s) 2+ Sn(s) + Mn+ (aq)
-
Falconi Academy Surplus had 55,000 shares of common stock and 5,000 shares of 1%, $10 par value preferred stock outstanding through December 31, 2016. Income from continuing operations for 2016 was...
-
Explain how due process is applied to agency actions and whether or not you believe it is fair that some decisions by agencies are not reviewable by an Article III court.
-
Tamar gives an intricately carved walking staff with a fair market value of $1,100 to Planned Parenthood, a public charity. What items would be sufficient to document this gift?
-
The way in which Aboriginal and/or Torres Strait Islander people and communities engage with community services will be vastly different to other communities. Discuss the impact of our individual...
-
Could you please provide 5 appropriate English case laws that I can use to answer the following: What is intoxication in law and how can a person avoid a contract entered into under it?
-
Alarmed by increasing rates of diabetes, heart disease, and cancer, Congress recently passed the Eat Your Vegetables Act of 2017. The EVYA has one provision: All Americans over the age of 21 must...
-
Imagine that you work as a sales associate for your local Gap store. The work schedule is always set a month in advance, but you want more flexibility. You ask the four other sales associates in the...
-
Suppose that A is an m n matrix with linearly independent columns and the linear system LS(A, b) is consistent. Show that this system has a unique solution.
-
What would the pattern look like for a laserbeam diffracted by the three crossed gratings of Fig. P.13.39? Fig. P.13.39?
-
Make a rough sketch of the Fraunhofer diffraction pattern that would arise if a transparency of Fig. P.13.40a served as the object. How would you filter it to get Fig. P.13.40b? Fig. P.13.40a and...
-
Repeat the previous problem using Fig. P.13.41 instead. previous problem Make a rough sketch of the Fraunhofer diffraction pattern that would arise if a transparency of Fig. P.13.40a served as the...
-
2-38 Cost of goods manufactured, income statement, manufacturing company. Consider the following account balances (in thousands) for the Peterson Company: Peterson Company Direct materials inventory...
-
A company provided the following data: Sales Variable costs Fixed costs Expected production and sales in units $600,000 380,000 150,000 45,000 units How much sales in dollars is necessary to generate...
-
On September 1, Hydra purchased $13,300 of inventory items on credit with the terms 1/15, net 30, FOB destination. Freight charges were $280. Payment for the purchase was made on September 18....

Study smarter with the SolutionInn App


