Balance each chemical equation. a. NaS(aq) + Cu(NO3)2(aq) b. NH(1) NH3(g) + N(g) c. HCl(aq) + O(g)
Question:
Balance each chemical equation.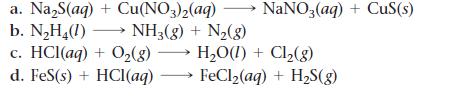
Transcribed Image Text:
a. Na₂S(aq) + Cu(NO3)2(aq) b. N₂H₂(1)→→→ NH3(g) + N₂(g) c. HCl(aq) + O₂(g) → H₂O(1) + Cl₂(g) d. FeS(s) + HCl(aq) FeCl₂(aq) + H₂S(g) NaNO3(aq) + CuS(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a NaSaq CuNO32aq NaNO3aq CuSs Balanced equation 2NaSaq ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Exercises 1-3, balance the chemical equation for each reaction.
-
The scatterplot shows the median weekly earning (by quarter) for men and women in the United States for the years from 2005 through 2017. The correlation is 0.974. a. Use the scatterplot to estimate...
-
Total current assets TOTAL ASSETS SHAREHOLDERS' EQUITY AND LIABILITIES: Shareholders' Equity: Preferred stock Common stock: Tk. 10 par 100,000 shares Share Premium 1690 2070 3060 3520 100 100 1000...
-
Transfer pricing is a significant area of concern for taxing authorities and multinational entities (MNE). Examine at least two (2) potential transfer pricing issues that create concern for both...
-
What is the yield to maturity for the bond issued by Xenon Inc.?
-
SuMar Company purchased a new piece of machinery by paying $2,000 down and agreeing to pay $1,000 at the end of each year for five years. The appropriate interest rate is 8%. Required 1. What is the...
-
Find a form for a voluntary dismissal in a California court.
-
Code Churn is a common metric used to measure the efficiency and productivity of software engineers and computer programmers. It?s usually measured as the percentage of a programmer?s code that must...
-
Required information [The following information applies to the questions displayed below] Caiman Distribution Partners is the Brazilian distribution company of a U.S. consumer products firm....
-
Consider the December transactions for Crystal Clear Cleaning that were presented in Chapter 5. Crystal Clear uses the perpetual inventory system. Dec. 2 Purchased 475 units of inventory for $2,850...
-
Consider the unbalanced equation for the combustion of hexane: Balance the equation and determine how many moles of O 2 are required to react completely with 7.2 moles of C 6 H 14 . C6H14(g) + O(8)...
-
Write the balanced chemical equation for the reaction of aqueous potassium hydroxide with aqueous iron(III) chloride to form solid iron(III) hydroxide and aqueous potassium chloride.
-
Perform the indicated operations in Problems 3948. 143 15 + + 210 124 11 1,085
-
Fill in the blank: The probability that a fair coin lands heads is 0.5. Therefore, we can be sure that if we toss a coin repeatedly, the proportion of times it lands heads will...
-
How may physical controls be used for these two classes of transactions?
-
How do brands work to create brand knowledge and influence purchase decisions?
-
What are the differences between customer journey maps, service blueprints, and customer experience maps?
-
What are some examples of presence cues? How are presence cues different online or in a physical store?
-
On March 1, 2015, Collier Enterprises purchases a 100% interest in Robby Corporation for $480,000 cash. Robby Corporation applies push-down accounting principles to account for this acquisition....
-
Wal-Mart is the second largest retailer in the world. The data file on the disk holds monthly data on Wal-Marts revenue, along with several possibly related economic variables. a) Using computer...
-
Picric acid is a military explosive formed via the nitration of phenol under conditions that install three nitro groups. Draw the structure and provide an IUPAC name for picric acid.
-
In each case, identify the most likely position at which monobromination would occur. (a) (b) (c) (d) N.
-
Identify the carboxylic acid and the alcohol that are necessary in order to make each of the following compounds via a Fischer esterification: a. b. c. CH 3 CH 2 CO 2 C (CH 3 ) 3
-
Supply chains are integral systems for the production and distribution of any product. Discuss how supply chains operate within the industry sector where you work. What is a problem that you know of...
-
for the company Sedgwick What diagnostic model(s) might most closely relate to the organizational problem? Why? How will data about the problem be collected? One could use processes, statistics,...
-
Officer Smith was responding to a traffic accident with injuries with his lights and sirens on. As he is going through an intersection he collides with another vehicle never making it to the original...

Study smarter with the SolutionInn App


