Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice.
Question:
Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice.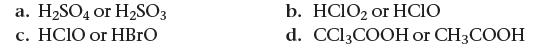
Transcribed Image Text:
a. H₂SO4 or H₂SO3 c. HCIO or HBrO b. HClO₂ or HCIO d. CC13COOH or CH3COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a HSO4 more oxygen atoms bonded ...View the full answer

Answered By

Emily Grace
With over a decade of experience providing top-notch study assistance to students globally, I am dedicated to ensuring their academic success. My passion is to deliver original, high-quality assignments with fast turnaround times, always striving to exceed their expectations.
4.90+
3+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Based on their molecular structure, pick the stronger acid from each pair of binary acids. Explain your choice. a. HF and HCI b. HO or HF c. HSe or HS
-
Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice. MISSED THIS? Read Section 17.10 a. HSO4 or HSO3 c. HCIO or HBrO b. HClO or HCIO d. CCI,COOH...
-
Sam's Insurance must choose between two types of printers. Both printers meet the firm's quality standard. Printer A costs $3,500 and is expected to last 3 years with operating costs of $380 per...
-
For the transfer function below, find the constraints on K 1 and K 2 such that the function will have only two j poles. K1s + K2 T(s) = s4 + K1s + s? + K2s + 1
-
Consider the following conversation between Keri Swasey, manager of a division that produces riding lawn mowers, and her controller, Stoney Lawson, a CMA and CPA: Keri: Stoney, we have a real...
-
Modify the diagram shown in Figure to include feed forward command compensation with a constant compensator gain Kf. Determine whether such compensation can eliminate steady-state error for step and...
-
How is a single plantwide factory overhead rate calculated?
-
Journalize the following transactions for Santa Fe Art Gift Shop. Explanations are not required. Feb. 3 Purchased $2,800 of merchandise inventory on account under terms 3/10, n/EOM and FOB shipping...
-
Overhead is applied on the basis of direct labour cost. During the first quarter of the year, the following activity took place in each of the accounts lists: Work in Process Bal. 17,000I Complete...
-
Based on molecular structure, arrange the oxyacids in order of increasing acid strength. Explain your choice. HClO3, HIO3, HBrO3
-
Based on molecular structure, arrange the binary compounds in order of increasing acid strength. Explain your choice. HTe, HI, HS, NaH
-
Verify that both the pressure and the vorticity field satisfy the Laplace equation for Stokes flow. Verify that the velocity field satisfies the biharmonic equation \[\begin{equation*}abla^{4}...
-
Question 3 (20 marks) Project overrun relates to a situation that occurs when the project's actual cost exceeds the initial budget. This causes a deficit in the project's financial needs and can slow...
-
Shares in XYZ Ltd were sold on 1/10/2017 for $45000. These shares were purchased for long term investment purposes on 31/10/1998 for $41500. In this case, how much do you have to pay in tax return?...
-
What procedure might be used to estimate the parameters of an ARMA model? Explain, briefly, how such a procedure operates, and why OLS is not appropriate.
-
The Bushrod Industrial Complex (BIC) employs about 60,000 salaried employees and another 4,000 as contractual staff. The salaried employees are paid once a month while the contract employees are paid...
-
discuss the role of stem cells in tissue regeneration and repair, including their potential therapeutic applications in regenerative medicine and the challenges associated with harnessing their...
-
Using the regression table in Exercise 14, answer the following questions. a) How was the t-ratio of 126.7 found for Paid Attendance? (Show what is computed using numbers found in the table.) b) How...
-
Burberrys competitive advantage is through its differentiation strategy. What risk should Burberry remain aware of?
-
Using Eq. (6.2), derive an expression for the focal length of a homogeneous transparent sphere of radius R. Locate its principal points. (n 1)d (n 1) R1 (6.2) R2 nRR2
-
A spherical glass bottle 20 cm in diameter with walls that are negligibly thin is filled with water. The bottle is sitting on the back seat of a car on a nice sunny day. Whats its focal length?
-
A thick glass lens of index 1.50 has radii of +23 cm and +20 cm, so that both vertices are to the left of the corresponding centers of curvature. Given that the thickness is 9.0 cm, find the focal...
-
can i have the answer as doc or typed please
-
Landmark Coal operates a mine. During July, the company obtained 500 tons of ore, which yielded 250 pounds of gold and63,100 pounds of copper. The joint cost related to the operation was $500,000....
-
Question 1: Compose an email report to the Accountant, providing an overview of the current status of Accounts Receivable and Accounts Payable. Ensure that your report adheres to the Policies and...

Study smarter with the SolutionInn App


