Calculate the solubility of Zn(OH) 2 (s) in 2.0 M NaOH solution. You must take into account
Question:
Calculate the solubility of Zn(OH)2(s) in 2.0 M NaOH solution.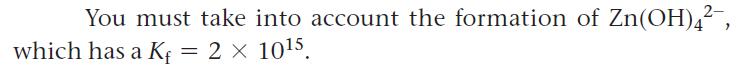
Transcribed Image Text:
You must take into account the formation of Zn(OH)4², 2 x 10¹5. which has a Kf =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the solubility of ZnOH2s in 20 M NaOH solutionwe must take into account the formation o...View the full answer

Answered By

ABHISHEK GAUTAM
I have an experience for teaching of two years and also i was working on chegg india for last 6 months as SME(subject matter expert) in mechanical engg. subject.I have a great passion for study and teaching .I want to make my career in teaching line itself.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following income statements illustrate different cost structures for two competing companies: Number of customers (a) Sales revenue (a x $230) Variable cost (ax $180) Variable cost (a x $0)...
-
A rigid bar ABCD is being supported by a hinge at A and two cable CE and DE. A measuring gauge determined that the vertical deflection at the free end of the bar is [y] mm. Consider these values: H =...
-
Harvatin Group reported net income totaling $1,000,000 for the year 2006. The following is additional information obtained from the Harvatin Group's financial reports: The Company purchased 100,000...
-
The two roots of a quadratic equation ax 2 + bx + c = 0 can be obtained using the following formula: b 2 - 4ac is called the discriminant of the quadratic equation. If it is positive, the equation...
-
Using 2008 as the base year, prepare a trend analysis of the following data, and tell whether the situation shown by trends is favorable or unfavorable. 2012 20 2010 2009 2008 Net sales Cost of goods...
-
Compare and contrast the arms-length or adversarial approach to the partnership approach to building customer-supplier relationships.
-
Shiloh supplies equipment to the automotive and commercial vehicle markets and other industrial customers. It specializes in materials and designs that reduce vehicle weight and increase fuel...
-
1. The Woody Company manufactures slippers and sells them at $10 a pair. Variable manufacturing cost is $4.50 a pair, and allocated fixed manufacturing cost is $1.50 a pair. It has enough idle...
-
Levinn's utility function is expressed as the following: U= C1 C2 0.3 where C1 is his first period consumption and C2 is his second period consumption. His income in the first period is $2500 and...
-
Explain why [Ni(NH 3 ) 4 ] 2+ is paramagnetic, while [Ni(CN) 4 ] 2- is diamagnetic.
-
Sulfide (S 2- ) salts are notoriously insoluble in aqueous solution. a. Calculate the molar solubility of nickel(II) sulfide in water. Ksp(NiS) = 3 x 10-16 b. Nickel(II) ions form a complex ion in...
-
Use grouping to factor the polynomial. + + + 3x2 + 2x + 6
-
Find the product. (3x+5y)
-
Are cryptocurrencies a foreign currency, financial instrument, or some other type of asset? How are cryptocurrencies accounted for under US GAAP? The FASB has proposed new guidance for accounting for...
-
What employee benefit plan records need to be retained permanently? Explain Why.
-
Merrill Lynch allows clients to view statements, make payments, and conduct stock trades, all online. Additionally, clients can read about market trends and other activities in the stock market....
-
Time Value of Money 1. A company needs to have $2,500,000 6 years from now for a building project. In an investment opportunity that pays 9%, how much will have to be deposited today in order to...
-
Banner Inc. bases its variable overhead performance report on the actual direct labor hours of the period. Data concerning the most recent year that ended on December 31 are as follows: Budgeted...
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
In a popular demonstration of induced emf, a lightbulb is connected across a large inductor in an RL circuit, as shown in Fig. 27.37. When the switch is opened, the bulb flashes brightly and may even...
-
You have a fixed length of wire to wind into an inductor. Will you get more inductance if you wind a short coil with large diameter, or a long coil with small diameter?
-
A car battery has a 12-V emf, yet energy from the battery provides the 30,000-V spark that ignites the gasoline. How is this possible?
-
For this video, please answer the following questions. Your perspective should be that of a voter within the state of Texas. How does each candidate present himself in this debate? For example, you...
-
The judge knows relatively little about each case. Group of answer choices True False
-
Attempt a fundamental evaluation of Torrington on a standalone basis

Study smarter with the SolutionInn App


