Closely examine the structure of glucose shown here. Does glucose exhibit optical isomerism (discussed in Section 22.3)?
Question:
Closely examine the structure of glucose shown here. Does glucose exhibit optical isomerism (discussed in Section 22.3)? If so, which carbon atoms are chiral?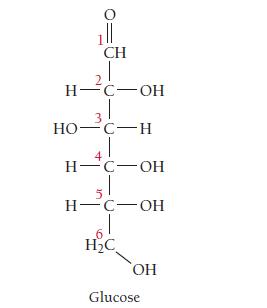
Transcribed Image Text:
1ll CH H-C-OH 3 HO-C-H H-C-OH H-C-OH H2C OH Glucose
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Any carbon atom with four different substituents att...View the full answer

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Examine the structure of deoxyribose, part of the backbone in DNA. Does deoxyribose exhibit optical isomerism? If so, which carbon atoms are chiral? - - C-
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Examine the structure of the following experimental design. Determine which of the three designs presented in the chapter would be most likely to characterize this structure. Discuss the variables...
-
Let S be the portion of the cylinder y = e x in the first octant that projects parallel to the x-axis onto the rectangle R yz : 1 y 2, 0 z 1 in the yz-plane. Let n be the unit vector normal to S...
-
Prepare a bank reconciliation from the following information: a. Balance per bank statement as of June 30, $4,862.77 b. Balance per books as of June 30, $2,479.48 c. Deposits in transit, $654.24 d....
-
Use the same information in Cornerstone Exercise 7-17, except that Irons uses the double-declining-balance method of depreciation. Required: Prepare the journal entry to record depreciation expense...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the ERR for this project. b. Is this project economically attractive? EOY 0 2 3 4 5 6 NCF -$100 $15 $15 $15...
-
The Town of Frostbite self-insures for some of its liability claims and purchases insurance for others. In an effort to consolidate its risk management activities, the Town recently decided to...
-
as Suppose X is a normal variable with mean and variance 2. Let f(x) be the density function of X. For a constant 0, define the tilted density function fo(x) = f(x)ex Mo where Me is the normalization...
-
Which compound is a lipid? a) HO b) c) CH H C I - OH OH H3C-(CH2)12 0 || OH 1 H2N - C - C-OH T CH T CH I S I CH3 OH d) H3C-CH2-0-CH2-CH3
-
Which compound is most likely to have a foul odor? a) b) CH3CH, NH, c) CH3-CH-0-CH3 d) CH3-CH-CH-OH CH3-C-0-CH3
-
Why would a business use harvest pricing? Why do many businesses using harvest pricing never exit the market?
-
Summarize your recommended costing approach and discuss how it will help Kay determine more accurate product costs for pricing different types of orders. What specific steps would you take to...
-
6. Katie sold the following assets during 2022. What depreciation rate can Jen take on each asset in 2022? (0.5 pts each, 1 pt total) Asset Convention Depreciation Rate? Half-year Mid-quarter...
-
Identify the reason(s) why net income must be adjusted for the noncash items on the statement of cash flows .? The accrual basis of accounting is used when recording transactions and preparing the...
-
Read the Managerial Problem stated at the beginning of the chapter 2 and the Managerial Solution at the end of the chapter 2, and then evaluate the following question: What sales and income tax rates...
-
When you bought some common stock of Guernsey Manufacturing Co., the price per share was $30.00 per share. After receiving a $0.33 per share dividend, you sold your shares at $36.00 per share. What...
-
STL Entertainment is considering the acquisition of a sight-seeing boat for summer tours along the Mississippi River. The following information is available: Cost of boat $500,000 Service life 10...
-
One of the significant and relevant accounts for this cycle is equipment. For this account, what would typically be the most relevant assertions for the auditor to consider? Why is it important for...
-
A 6.8C charge and a -4.7C charge are inside an uncharged sphere. Whats the electric flux through the sphere?
-
A 2.6C charge is at the center of a cube 7.5 cm on each side. Whats the electric flux through one face of the cube? (Think about symmetry, and dont do an integral.).
-
The electric field at the surface of a 5.0-cm-radius uniformly charged sphere is 90 kN/C. Whats the field strength 10 cm from the surface?
-
Anne purchased shares on 1 December 1985 for $1,000. She sold the shares on 1 December 2019 for $1,500. What is the minimum assessable capital gain on the shares to be included in her tax return for...
-
Solo Corp started operations on August 1, 2023, and is a calendar year corporation. It incurred 32,000 in organizational expenses that year. What amount is deductible on Solo Corp's tax return in...
-
A furniture store is promoting a no HST (14%) event for the Fall Sale. Consider a sofa that costs $ 985 (HST included). What should the price of the sofa be during the Fall Sale? Show your...

Study smarter with the SolutionInn App


