Complete and balance each combustion reaction equation. a. S(s) + O(g) C. Ca(s) + O(g) b. C3H6(g)
Question:
Complete and balance each combustion reaction equation.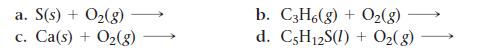
Transcribed Image Text:
a. S(s) + O₂(g) C. Ca(s) + O₂(g) b. C3H6(g) + O₂(g) d. C5H12S(I) + O₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Ss O8 b 2 C3H6g 9 ...View the full answer

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete and balance each combustion equation. a. C4H9OH + O2 ( ? b. CH3NO2 + O2 ( ?
-
Complete and balance each combustion equation. a. B2H6 + O2 ( ? (The oxide of boron formed is B2O3.) b. Al2S3 + O2 ( ? (The oxide of sulfur formed is SO2.) c. Al2S3 + O2 ( ? (The oxide of sulfur...
-
Complete and balance each combustion reaction equation: a. C4H6(g) + O(g) C. CS(s) + O(g) b. C(s) + O(g) d. C3H8O(l) + O(g)
-
The Carter Caterer Company must have the following number of clean napkins available at the beginning of each of the next four days: day 1, 1500; day 2, 1200; day 3, 1800; day 4, 600. After being...
-
Consider the following abbreviated financial statements for Cabo Wabo, Inc.: a. What is owners' equity for 2007 and 2008? b. What is the change in net working capital for 2008? c. In 2008, Cabo Wabo...
-
Starbucks dominates the coffee roasting, distribution, and pouring market. Starbucks owns or licenses over ten thousand stores internationally that sell primarily various coffee-related drinks...
-
The thermal decomposition of limestone takes place as \[ \mathrm{CaCO}_{3}(\mathrm{~s}) ightleftharpoons \mathrm{CaO}(\mathrm{s})+\mathrm{CO}_{2}(\mathrm{~g}) \] Calculate the decomposition pressure...
-
H & K Electronic Warehouse sells a 12-pack of AAA batteries, and this is a very popular item. Demand for this is normally distributed, with an average of 50 packs per day and a standard deviation of...
-
Bridge City Consulting bought a building and the land on which it is located for $205,000 cash. The land is estimated to represent 50 percent of the purchase price. The company paid $10,000 for...
-
Lombard Ltd has been offered a contract for which there is available production capacity. The contract is for 20,000 identical items, manufactured by an intricate assembly operation, to be produced...
-
Write a balanced chemical equation for the reaction of solid strontium with iodine gas.
-
What volume of a 1.50 M HCl solution should you use to prepare 2.00 L of a 0.100 M HCl solution? a) 0.300 L b) 0.133 L c) 30.0 L d) 2.00 L
-
STL Inc. reported 2009 earnings per share of $3.20 and had no extraordinary items. In 2010, EPS on income before extraordinary items was $2.99, and EPS on net income was $3.49. Is this a favorable...
-
11 Determine the retail price of a child's toy that cost $1.69 and has a 45 1/2% markup. Round to 2 decimal points. Edit View Insert Format Tools Table
-
1. Suppose that the auto loans from a bank are normally distributed with a mean of $23,334 and a standard deviation of 3,412. What is the probability that a randomly selected loan will be for more...
-
19. What is the beta for a portfolio equally weighted in four assets: A with beta =.9; asset B, which has half the risk of the market; C, which has beta=1.5; and D, with beta-1.2?
-
A loan officer compares the interest rates for 48-month fixed-rate auto loans and 48- month variable-rate auto loans. Two independent, random samples of auto loan rates are selected. The resulting...
-
29. E-Loan, an online lending service, offers a 36-month auto loan at 7.56% compounded monthly to applicants with good credit ratings. If you have a good credit rating and can afford
-
Explain the difference between an informational objective and a motivational objective.
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Which of the following compounds would you expect to be most acidic? Justify your choice.
-
Identify which of the following compounds is expected to be a stronger base. Justify your choice. N. N.
-
Predict the product of the following reaction, and propose a mechanism for its formation. Na, CH,OH NH;
-
A bird is flying south at a rate of 45 miles per hour while being pushed east by wind with a speed of 12 miles per hour. What is the magnitude of the bird's resultant vector?
-
Price Corporation acquired 100 percent ownership of Saver Company on January 1, 20X8, for $168,000. At that date, the fair value of Saver's buildings and equipment was $36,000 more than the book...
-
Watch https://youtu.be/U3MtvvNjUR4 What do you think of Dr. Saint's ideas about barriers to change? What do you think about social learning? Could this tool be used to make real change? How can the...

Study smarter with the SolutionInn App


