Consider the generic chemical equation A + 3B 2C. Let circles represent molecules of A and
Question:
Consider the generic chemical equation A + 3B → 2C.
Let circles represent molecules of A and squares represent molecules of B. The diagram shown at the far right represents the amount of B available for reaction. Which diagram in the answer options accurately represents the amount of A necessary to completely react with B?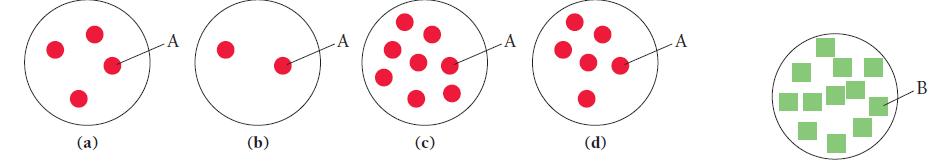
Transcribed Image Text:
(a) A (b) -A (c) A (d) A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Since the balanced equation indic...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Under certain conditions, sodium reacts with oxygen to form sodium oxide according to the reaction: A flask contains the amount of oxygen represented by the diagram shown at far right. Which of the...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Assume the premises: S. S-P. P~N, ~R-N. Give a detailed informal proof of: R. Number each step and give an explanation for each. Write all the steps in the proof at once in the Math Editor. (it works...
-
Data for Ulrich Company are presented in P12-9B. Further analysis reveals that accounts payable pertain to merchandise creditors. Instructions Prepare a statement of cash flows for Ulrich Company...
-
A solar collector consists of a parallel plate channel that is connected to a water storage plenum at the bottom and to a heat sink at the top. The channel is inclined 0 = 30 from the vertical and...
-
Based on the following pedigree for a trait determined by a single gene (affected individuals are shown as filled symbols), state whether it would be possible for the trait to be inherited in each of...
-
Paige Corporation makes a mechanical stuffed alligator that sings the Martian national anthem. The following information is available for Paige Corporations anticipated annual volume of 500,000...
-
When a company lends money to employees at a rate of 4%, the company will record ______. Multiple choice question. a liability called Accounts Payable a liability called Notes Payable an asset called...
-
Would a grant of 24,000 by the state to each district be equalizing in its effect on the two districts? 2. Would a state grant of $100 per student be equalizing in its effect on the two districts?...
-
Sodium and chlorine react to form sodium chloride: What is the theoretical yield of sodium chloride for the reaction of 55.0 g Na with 67.2 g Cl 2 ? a) 1.40 * 10 2 g NaCl b) 111 g NaCl c) 55.4 g NaCl...
-
Identify the reactants and products in this chemical equation. 4 NH 3 (g) + 5 O2( g) 4 NO(g) + 6 H 2 O(g)
-
Some people might recall being told to be sure to put on a hat when outside in cold weather because "you lose all the heat out the top of your head." Comment on the validity of this statement.
-
What are information systems? Give three examples of IT-related internal controls What is the auditor's responsibility when evaluating IT systems? Explain the difference between operating software...
-
1. Brief history of Kmart 2. Kmart leaders made bad decisions: 2a. What bad decision was made that brought the company into trouble? 2b. What could the company have done to protect itself from these...
-
What distinguishes maritime law from other areas of law? Are there any subdivisions within maritime law? If so, what are they, and how are they distinguished from each other? What are some of the...
-
Theory and Fundamentals of Radar Operations. Explain the history of radar and the importance it has within the ATC system. How has it evolved to the equipment used by today's air traffic controllers?...
-
Terminology and Acronyms of the Different Accounting Institutions Instructions Identify the following acronyms referring to the accounting profession and explain the purpose of each of them. 1. FASB...
-
Identify the errors in the following income statement and prepare a corrected incomestatement: CARLSBAD COMPANY Income Statement For the Year Ended February 29, 2012 Revenue from sales: $4,400,000...
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
The vapor pressure of liquid benzene is 20,170 Pa at 298.15 K, and Î H vaporization =30.72 kJ mol -1 at 1 atm pressure. Calculate the normal and standard boiling points. Does your result for...
-
Benzene(l) has a vapor pressure of 0.1269 bar at 298.15 K and an enthalpy of vaporization of 30.72 kJmol 1 . The C P,m of the vapor and liquid phases at that temperature are 82.4 and 136.0 J K 1 mol...
-
Use the values for G o f (CCl 4 , l) and G o f (CCl 4 , g) from Appendix B to calculate the vapor pressure of CCl 4 at 298.15 K.
-
Make a Relational Model Table and Define Keys from Entity Relation Diagram Q2: Design the World Music Association Database using given ERD. ORCHESTRA "Orchestra Name City Country Music Director...
-
Discuss the business benefits of adopting the relational database model within an organization. How does a relational model create or enhance business value? Are there any limitations to using a...
-
. Do policies exist within the Linux workstation or server environment? If so, what are they? . Are there ways that Windows policies can be leveraged to enhance corporate or security policy?

Study smarter with the SolutionInn App


