Consider the reaction: A reaction mixture at 100 C initially contains [NO 2 ] = 0.100 M.
Question:
Consider the reaction: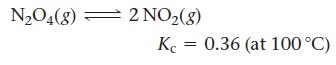
A reaction mixture at 100 °C initially contains [NO2] = 0.100 M. Find the equilibrium concentrations of NO2 and N2O4 at this temperature.
Transcribed Image Text:
N₂O4(8) = 2 NO₂(8) Kc 0.36 (at 100 °C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
NO48 2 NOg Initial Change Equil Qc NO N04 N04 NO 000 0100 0100 000 00 Q ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Mr. Creary has hired you to estimate a selling price for his house using the direct market comparison method. You have compiled the following information: Features Sale Price Sale Time Lot Size House...
-
Consider the reaction: 2 H 2 S (g) 2 H 2 (g) + S 2 (g) Kc = 1.67 10 -7 A reaction mixture initially contains [H 2 S] = 0.010 M. Find the equilibrium concentrations of H 2 and S 2 .
-
Player 1 and Player 2 are involved in a dispute. Player 2 is either Weak or Strong. Player 2's strength is private information. 2 knows his strength; 1 knows that 2 is Strong with probability p=0.35...
-
A project is at the multi-functional planning phase. The project manager realizes that producing a complex application is not feasible with the current number of resources and decides to hire more...
-
What is a focused value stream?
-
In each cycle, an engine removes 150 J from a reservoir at 100 o C and gives off 125 J to a reservoir at 20oC. (a) What is the efficiency of this engine? (b) What is the ratio of its efficiency to...
-
True or False: If \(P W>0\), then \(I R R>M A R R\).
-
The following T-accounts represent November activity. Additional Data Materials of $113,600 were purchased during the month, and the balance in the Materials Inventory account increased by $11,000. ...
-
On the first day of their vacation the Morales family drove 312 miles in 6 hours at that rate how far will they travel the next day if they drive for 8 hours
-
Does the value of the equilibrium constant depend on the initial concentrations of the reactants and products? Do the equilibrium concentrations of the reactants and products depend on their initial...
-
For the reaction, A(g) 2 B( g), K c = 4.0. A reaction mixture at equilibrium contains [A] = 1.0 M. What is the concentration of B in the reaction mixture? (a) 0.50 M (b) 1.0 M (c) 2.0 M (d) 4.0 M
-
Evaluate the decision by IBM to consolidate all of its worldwide advertising with the Ogilvy & Mather agency. What are the pros and cons of using one agency to handle all of its advertising?
-
On most small to medium-sized projects, how many intermediate milestones should be identified in the charter?
-
Risks that have been identified and may or may not happen are referred to as known unknowns, and a _______________ should be established to cover them if they are triggered. a. contingency reserve b....
-
What are organizational values, and why should a project manager be aware of them?
-
When is the best time to on-board core team members?
-
For what five activities is the project steering team responsible?
-
This year Bertrand Company sold 40,000 units of its only product for $ 25 per unit. Manufacturing and selling the product required $ 200,000 of fixed manufacturing costs and $ 325,000 of fixed...
-
Suppose that the laptop of Prob. 2.16 is placed in an insulating briefcase with a fully charged battery, but it does not go into sleep mode, and the battery discharges as if the laptop were in use....
-
What is the basic distinction between the scientific method and other ways of looking at the natural world?
-
Scientific models do not correspond exactly to reality. Why are they nevertheless so useful?
-
What does a year correspond to in terms of observations of the sun and stars?
-
Requirement 2. Prepare the income statement for the year ended March 31, 2022. (Click to view the information you will need to complete the income statement. Robin Sporting Goods Company Income...
-
Marketing Study: will be able to construct an academic study linked to a contemporary marketing topic, issue, or trend. Students will be required to work either individually or in groups of 2...
-
At January 1, 2024, Crane Corporation, a public company, reported the following property, plant, and equipment accounts: Accumulated depreciation-buildings $30,200,000 Accumulated...

Study smarter with the SolutionInn App


