Consider the reaction: If a solution initially contains 0.210 M HC 2 H 3 O 2 ,
Question:
Consider the reaction:
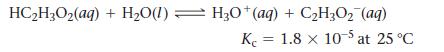
If a solution initially contains 0.210 M HC2H3O2, what is the equilibrium concentration of H3O+ at 25 °C?
Transcribed Image Text:
HC₂H3O₂ (aq) + H₂O(1) H3O+(aq) + C₂H3O₂ (aq) K 1.8 x 10-5 at 25 °C =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
19...View the full answer

Answered By

Navashree Ghosh
I believe in quality work and customer satisfaction. So, I can assure you that you will get quality work from me when you hire me. Let's work together and build a long-term association.
4.90+
82+ Reviews
116+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A chemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. The drug molecules bind the protein in a...
-
Compare and contrast Taylorism, Theory of Constraint, and Toyota Production System
-
The solution containing no added KNO3 for Figure 7-1 contains 5.0 mM Fe(NO3)3, 5.0 M NaSCN, and 15 mM HNO3. We will use Davies activity coefficients to find the concentrations of all species in the...
-
A ball is attached to one end of a wire, the other end being fastened to the ceiling. The wire is held horizontal, and the ball is released from rest (see the drawing). It swings downward and strikes...
-
What is the purpose of a base period?
-
Some early computers protected the operating system by placing it in a memory partition that could not be modified by either the user job or the operating system itself. Describe two difficulties...
-
What is an Enabling System (Supplier) role?
-
1. A factor that causes or leads to a change in a cost or activity is a (n) a. Driver. b. Intercept. c. Slope. d. Variable term. e. Cost object. 2. Which of the following would probably be a variable...
-
You plan to migrate an on-premises Apache Spark deployment to a managed Apache Spark service in Azure. To which Azure service can you migrate the on-premises deployment?
-
Consider the reaction: If a reaction mixture initially contains 0.175 M SO 2 Cl 2 , what is the equilibrium concentration of Cl 2 at 227 C? SOCl(g) = SO(g) + Cl(g) K 2.99 x 10-7 at 227 C =
-
For the reaction shown here, K c = 255 at 1000 K. CO(g) + Cl 2 (g) COCl 2 (g) If a reaction mixture initially contains a CO concentration of 0.1500 M and a Cl 2 concentration of 0.175 M at 1000 K,...
-
A new television series is to be shown. A broadcasting executive feels that his uncertainty about the rating that the show will receive in its first month can be represented by a normal distribution...
-
A van traveling at a speed of 3 8 . 0 mi / h needs a minimum of 5 0 . 0 ft to stop. If the same van is traveling 6 6 . 0 mi / h , determine its minimum stopping distance ( in ft ) , assuming the same...
-
A n aircraft of weight 3 4 7 0 0 0 N is catapulted off the deck of an aircraft carrier. if the aircraft starts at rest, and reaches a velocity of 7 2 m / s 2 in 2 . 5 seconds, how much force is...
-
After you read the case study below, there are 11 releases of information situations that transpired this day. Read the Situation and the Action and then comment on whether you agree or disagree with...
-
When Great Britain voted to leave the eurozone, the pound depreciated 17% against the dollar. It also raised fears that the eurozone, which uses the euro as a common currency, would fall apart....
-
As a vice president for Pharmacia (which was acquired by Pfizer in 2003), Dr. Peter Rost was in charge of worldwide marketing for the drug Genotropin, which is a synthetic human growth hormone that...
-
Munger.Com began operations on January 1, 2006. The company reports the following information about its investments at December 31, 2006: Required: a. Show how each of these investments are reported...
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
A pipeline is needed to transport medium fuel oil at 77F. The pipeline needs to traverse 80 mi in total, and the initial proposal is to space pumping stations 2 mi apart. The line needs to carry 750...
-
Soccer on a windy day. A professional soccer player can kick a ball with a speed of about 30 m/s (about 75 mi/h). Does air drag play a significant role in the trajectory of the ball? Give a reason...
-
When airplanes land or take off, they always travel along a runway in the direction that is into the wind because the lift force on an airplane wing depends on the speed of the airplane relative to...
-
Flint Corp. s trial balance at December 3 1 , 2 0 2 3 is properly adjusted except for the income tax expense adjustment. Flint Corp. Trial Balance December 3 1 , 2 0 2 3 Dr . Cr . Cash $ 3 3 7 2 0 0...
-
On 1 July 2023, Bronzed Aussie Ltd sells a caravan to Cairns Ltd. The caravan has a normal sales price of $19 019. Rather than selling the item for its normal sales price, Bronzed Aussie Ltd sells...
-
On August 1 , 2 0 2 2 , Colombo Company s treasurer signed a note promising to pay $ 1 2 1 , 8 0 0 on December 3 1 , 2 0 2 2 . The proceeds of the note were $ 1 1 4 , 6 0 0 . Record the journal entry...

Study smarter with the SolutionInn App


