Determine the composition and phase present at points C and D on the CrFe phase diagram(see Problem
Question:
Determine the composition and phase present at points C and D on the Cr–Fe phase diagram (see Problem 35 for diagram).
Problem 35
Determine the composition and phases present at points A and B on the Cr–Fe phase diagram.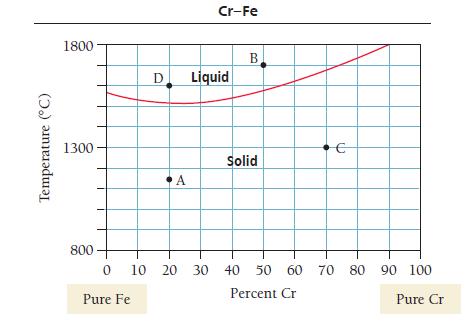
Transcribed Image Text:
Temperature (°C) 1800 1300 800 D Pure Fe A Cr-Fe Liquid B Solid € 0 10 20 30 40 50 60 70 80 90 100 Percent Cr Pure Cr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The CrFe phase diagram provides valuable insights into the composition and phases of the iro...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the curve C between the points (1, 0, -2) and (0, 1,-1) at the intersection of the cylinder around the z-axis with radius 1 and the plane 2x+y+z=0. Determine the line integral of the...
-
Determine the composition, relative amounts, and phases present at points C and D on the CoCu phase diagram (see Problem 37 for phase diagram). Problem 37 Determine the composition, relative amounts,...
-
Determine the composition and phase present at point A on Figure 25.5. Temperature 1500 1400 1300 1200- 1100 1000- 900 Liquid 1084 C A D Solid alloy C B. 1455 C 0 10 20 30 40 50 60 70 80 90 100 Mole...
-
Linda has d dollars in an account that pays 1.4% interest, compounded weekly. She withdraws w dollars. Express her first weeks interest algebraically.
-
Fuentez Mining Company purchased land containing an estimated 20 million tons of ore for a cost of $8,800,000. The land without the ore is estimated to be worth $1,600,000. The company expects that...
-
How much must you deposit at the end of each quarter in an account that pays a nominal interest rate of 20 percent, compounded quarterly, if at the end of five years you want $10,000 in the account?...
-
For the following products and countries, identify the type of warehouse that should be used as well as the method of transportation that should deliver the product to end users. Using the Internet,...
-
Refinancing of Short-Term Debt On December 31, 2010, Santana Company has $7,000,000 of short-term debt in the form of notes payable to Golden State Bank due in 2011. On January 28, 2011, Santana...
-
Suppose carpet for a 12 ft by 10 ft room costs $400. Find the cost to carpet a room 36 ft by 30 ft. The cost to carpet the room is $
-
The elements Mn and Si are added to steel to improve its properties in electric motors, and some C is often found as a detrimental impurity. Which of these elements fills interstitial holes in the Fe...
-
Determine the composition and phases present at points A and B on the CrFe phase diagram. Temperature (C) 1800 1300 800 D Pure Fe A Cr-Fe Liquid B Solid 0 10 20 30 40 50 60 70 80 90 100 Percent Cr...
-
A manufacturer of sprinkler systems designed for fire protection claims that the average activating temperature is at least 135F. To test this claim, you randomly select a sample of 32 systems and...
-
How does Countee Cullen's "Heritage" depict his conflict with American and African heritage?
-
Linda has been filing her individual tax returns on a calendar year basis for ten years. She wants to start a sole proprietorship on April 1st. When does her business tax year end?
-
Lambda Corporation has current liabilities of $432,000, a quick ratio of 1.9, inventory turnover of 4.6, and a current ratio of 3.3. What is the cost of goods sold for Lambda Corporation?
-
2. Cougar Gold Mining Corporation (CGMC) has 5 million shares of common stock outstanding and 200,000 bonds with a 6 percent coupon rate outstanding, par value $1,000 each. The common stock currently...
-
Controlling Program Flow w/i an ATM Using selection statements & loops to complete the following: Validate the user's PIN -- only allow them to attempt three (3) times before ending the program If...
-
Assume Victory uses the average-cost method to account for its process costing system. VICTORY COMPANY For the month of November: Number of units transferred to finished goods....... 700,000 Number...
-
Evaluate how many lines there are in a true rotational spectrum of CO molecules whose natural vibration frequency is w = 4.09 1014 s1 and moment of inertia I = 1.44 1039 g cm2.
-
A piece of wire with a constant cross-sectional area has a total resistance R 0 . The wire is cut into three pieces of equal length, which are then reconnected to form an equivalent resistor with...
-
You are given four capacitors, each with capacitance C. Devise two ways to connect these capacitors to get a total equivalent capacitance greater than C.
-
You are given four capacitors, each with capacitance C. Devise two ways to connect these capacitors to get a total capacitance less than C.
-
Let : [0,1] R be a continuous function. Let R have the usual Euclidean coordinates (x, y, z), and suppose (0) = (-10, 5,7) and f(1) = (10,3,3). Show that Im(f) intersects the plane in R given by x =...
-
Phew! Not too steep! Once inside the water tower, Zachary easily defeats the kidnapper in hand-to-hand combat, but getting the princess outside the tower will be tricky, since there is no ladder...
-
Write a program for pretty printing C++ programs; that is, for printing programs with consistent use of indentation, the number of spaces between tokens such as key words, parentheses, brackets,...

Study smarter with the SolutionInn App


