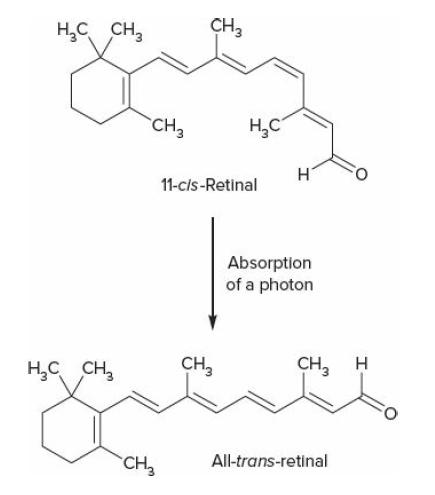
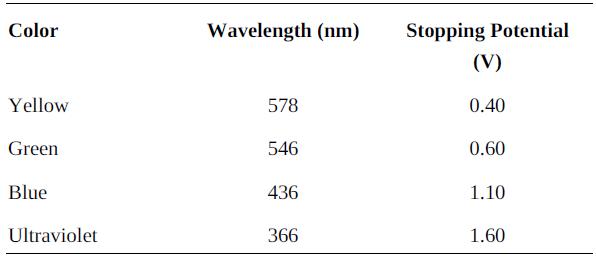
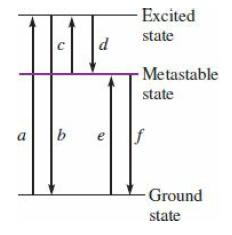
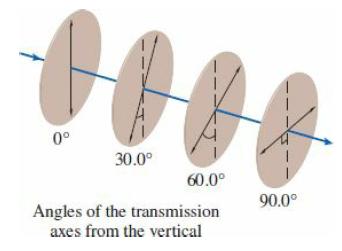
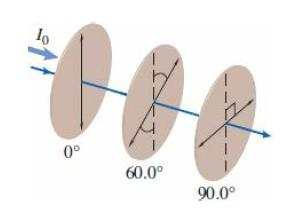
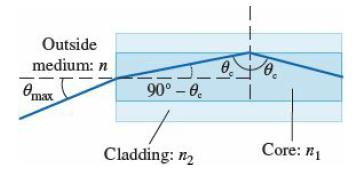
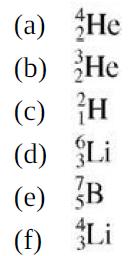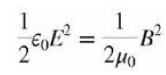College Physics With An Integrated Approach To Forces And Kinematics 5th Edition Alan Giambattista - Solutions
Discover comprehensive resources for "College Physics With An Integrated Approach To Forces And Kinematics 5th Edition" by Alan Giambattista. Access the online answers key, solution manual, and chapter solutions to enhance your understanding. Benefit from detailed step-by-step answers and solved problems to master complex concepts. Utilize the instructor manual and test bank for thorough preparation. Download the solutions PDF and explore questions and answers for each chapter. This textbook is your ultimate guide to physics, offering free downloads to aid your study. Achieve academic success with these invaluable resources at your fingertips.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()