Determine the product of the reaction: CH3 I CH3 -CH=C-CH3 + HBr CH3 T a) CHBr -CH=C-CH3
Question:
Determine the product of the reaction: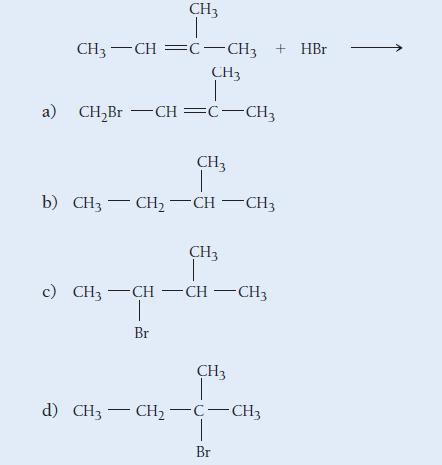
Transcribed Image Text:
CH3 I CH3 -CH=C-CH3 + HBr CH3 T a) CH₂Br -CH=C-CH3 CH3 b) CH3 - CH₂ -CH-CH3 CH3 c) CH3 -CH=CH-CH3 Br CH3 d) CH3 - CH₂ -C-CH3 Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
CH3 ...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Demonstrate your knowledge of palladium cross-coupling reactions by suggesting structures for compounds (14) and (15). Suggest reagent(s) that would facilitate the cross-coupling reaction between...
-
When (S)-1-bromo-1-fluoroethane reacts with sodium methoxide, an S N 2 reaction takes place in which the bromine atom is replaced by a methoxy group (OMe). The product of this reaction is...
-
Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce 24 Na. When removed from the reactor, the sample is radioactive, with activity of 2.54 10 4 dpm....
-
On July 31, 2023, Magnuss Muffins paid $18,000 for business insurance for the next year. Record the entries for the purchase of the insurance by recording it as an expense and then making a year-end...
-
Refer to CVS's financial statements in the Supplement to Chapter 5 and to the following data for Walgreens: 2008 2007 2006 Cost of goods sold Accounts payable Increase in merchandise iventories 412...
-
D. Deni commenced business on 1 April 2014 by depositing Sh.8 million into the business bank account. All receipts and payments were channelled through this business account. During the year ended 31...
-
What are the different types of consulting and litigation support activities for fraud and forensic accounting professionals?
-
In Part I of the case, you performed preliminary analytical procedures for Pinnacle. The purpose of Part II is to identify factors influencing risks and the relationship of risks to audit evidence....
-
Do the following insert and delete operations using linear probing method on an empty hash table with the size of 11. Then, find the average probing number on this hash table. SHOW your steps. Hash...
-
Determine the product of the reaction: CH3-CH-CH-OH CH3 a) CH3-CH - CH3 Cl b) CH3-CH - CH3 I CH3 CI c) CH3CCH, - OH CH3 d) CH3-CH-CH - Cl | CH3 + HCl
-
Determine the type of reaction (substitution, dehydration, oxidation, or reaction with an active metal) that occurs in each case, and write formulas for the products. CH3 (a) CH3-CH-CH-CH-OH + HBr...
-
Verify the conditional likelihood (4.29). k II i=1 1 1+ exp( d;)' (4.29)
-
What are prepaid expenses. ? What is accrued expenses ? How Commodities and services that have been purchased for consumption but are unconsumed at the end of the accounting period. Explain briefly
-
Why should the administrator secure information on budget needs from each department head? Explain briefly
-
What to data sources are used in the government wide determine equity tool?
-
Estimate your revenues and expenses, month by month, for the first year of your startup coffee shop using the details you previously entered in your business plan (such as price and production...
-
Identifying the major challenges that need to be addressed before self-driving trucks can be deployed. These challenges may include technical, regulatory, and societal issues that must be resolved...
-
Assume that you are considering two projects which are mutually exclusive. Project X3 will cost $400,000 and will return $200,000 per year for three years. Project Y6 will cost $300,000 and will...
-
The swap spread is the difference between the swap rate and the equivalent-maturity Treasury bond yield. Explain why a widening swap spread may be a signal of deteriorating economic conditions. Plot...
-
A net charge of 5.0 C is applied on one side of a solid metal sphere 2.0 cm in diameter. Once electrostatic equilibrium is reached, and assuming no other conductors or charges nearby, what are (a)...
-
A positive point charge q lies at the center of a spherical conducting shell carrying net charge 3/2 q. Sketch the field lines both inside and outside the shell, using eight field lines to represent...
-
A total charge of 18C is applied to a thin, square metal plate 75 cm on a side. Find the electric field strength near the plates surface.
-
Construct appropriate tables or graphs to accurately display the results of the data analysis in the case study Maruti Suzuki India: Defending Market Leadership''. Include explanations of the meaning...
-
The telephone reservation department for a major car rental firm has the daily shift requirements for operators below: Day Su M Tu W Th F Sa Operators 4 8 8 7 7 6 5. Prepare a weekly workshift...
-
What need to be learned and applied from the past experiences to the new phenomena of pure drinking water in Egyptian Villages?

Study smarter with the SolutionInn App


