Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce
Question:
Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce 24Na. When removed from the reactor, the sample is radioactive, with β activity of 2.54 × 104 dpm. The decrease in radioactivity over time was studied, producing the following data: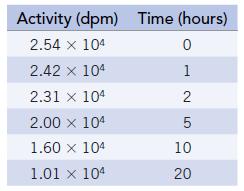
(a) Write equations for the neutron capture reaction and for the reaction in which the product of this reaction decays by β emission.
(b) Determine the half-life of sodium-24.
Transcribed Image Text:
Activity (dpm) Time (hours) 2.54 x 104 0 2.42 x 104 1 2.31 x 104 2 5 2.00 x 104 1.60 x 104 1.01 x 104 10 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a The neutron capture reaction that produces sodium24 can be repre...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Immanuel Kant believed that the results of a decision are not as important as the reason for making it. This is the principle behind Outilitarian ethics. O deontological ethics. Rawlsian justice. O...
-
Required a. Use professional judgment in deciding on the preliminary judgment about materiality for earnings, current assets, current liabilities, and total assets. Your conclusions should be stated...
-
Data A.1 on page 136 introduces a study that examines the effect of light at night on weight gain in a sample of 27 mice observed over a fourweek period. The mice who had a light on at night gained...
-
A 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2: At equilibrium the total pressure in the container is 1.300 atm. Find the values of Kp...
-
At a recent staff meeting, the management of Hom Technology Products was considering discontinuing the Hercules line of laptop computers from the product line. The chief financial analyst reported...
-
At sunset, red light travels horizontally through the doorway in the western wall of your beach cabin, and you observe the light on the eastern wall. What is the width of the central maximum in the...
-
What is life?
-
The following accounts appear in the ledger of Alexis Inc. after the books are closed at December 31. Common Stock, no par, $1 stated value, 400,000 shares authorized; 300,000 shares issued...
-
A hiker walks 27.0 km from her base camp at 35 south of east. The next day, she walks 41.0 km in a direction 65 north of east and discovers a forest
-
The average energy output of a good grade of coal is 2.6 10 7 kJ/ton. Fission of 1 mol of 235 U releases 2.1 10 10 kJ. Find the number of tons of coal needed to produce the same energy as 1 lb of...
-
A sample of wood from a Thracian chariot found in an excavation in Bulgaria has a 14 C activity of 11.2 dpm/g. Estimate the age of the chariot and the year it was made. (t for 14 C is 5.73 10 3...
-
Why are the screening activities so important in the selection of focus group participants? Develop a screening form that would allow you to select participants for a focus group on the benefits and...
-
Form a small group of four or five students and discuss your preference for the type of learning environment in which you can take a human resource management course for university credit and learn...
-
Visit the New Belgium Brewing Web site (www.newbelgium.com) to learn more about the company. The company has a strong culture that values environmental sustainability. Here are its core beliefs: We...
-
A company discovers that a poorly performing factory has a significant level of voluntary turnover compared to other locations. The company is not sure if it is a symptom of poor management or...
-
With your teammates, look at some of the online jobopening announcements. Consider at least a dozen ads and summarize those that include a description of social responsibility commitments or...
-
Label each of the following statements true, false, or uncertain. Explain briefly. a. US GDP was 38 times higher in 2018 than it was in 1960. b. When the unemployment rate is high, the participation...
-
The cost behavior patterns below are lettered A through H. The vertical axes of the graphs represent total dollars of expense, and the horizontal axes represent production in units, machine hours, or...
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
Compute the time required to reduce the depth in the tank shown in Fig. 6.14 by 225 mm if the original depth is 1.38 m. The tank diameter is 1.25 m and the orifice diameter is 25 mm. dh
-
Compute the time required to reduce the depth in the tank shown in Fig. 6.14 by 1.50 m if the original depth is 2.68 m. The tank diameter is 2.25 m and the orifice diameter is 50 mm. dh
-
Compute the time required to empty the tank shown in Fig. 6.14 if the original depth is 18.5 in. The tank diameter is 22.0 in and the orifice diameter is 0.50 in. dh
-
A 6% sales tax function applied to any price p can be described as follows: f (p) is 0.06p rounded to the nearest cent, where half-cents are rounded up. For example, 0.06 (1.25) = 0.075, so f (1.25)...
-
The following data are taken from the financial statements of Sandhill Co.. The data are in alphabetical order. Accounts payable $26,800 Net sales $534,000 Accounts receivable 69,000 Other current...
-
On March 1, Dortex Manufacturing Ltd. (Dortex) purchased a factory with a lot of land and a machine for $818,000. Dortex paid legal fees of $2,000 for this purchase, renovation costs on the building...

Study smarter with the SolutionInn App


