Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict
Question:
Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change.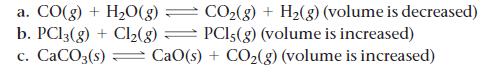
Transcribed Image Text:
CO₂(g) + H₂(g) (volume is decreased) PCI,(g) (volume is increased) a. CO(g) + H₂O(g) - - b. PC13(g) + Cl₂(g) c. CaCO3(s) CaO(s) + CO₂(g) (volume is increased)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a When the volume is decreased the reaction will try to offset the change by shifting in the directi...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume...
-
Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change. a. 1(g) 21(g)...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
1. A projectile is launched in a vertical plane, at an angle 0 with initial velocity vo. It must be caught in a frictionless circular tube of radius R in such a way that the trajectory of the...
-
Refer to Exercise 15-3. Suppose the following input prices are provided for each year: Required: 1. Compute the profit-linked productivity measure. By how much did profits increase due to...
-
Show by writing appropriate chemical equations how each of the following compounds could be converted to 3-hexyne: (a) 1-Butene (b) 1, 1-Dichlorobutane (c) Acetylene
-
Where and how do Users obtain system requirements for development?
-
Morro Bay Surfboards manufactures fiberglass surfboards. The standard cost of direct materials and direct manufacturing labor is $225 per board. This includes 30 pounds of direct materials, at the...
-
Pearl Products Limited of Shenzhen, China, manufactures and distributes toys throughout South East Asia. Three cubic centimeters ( cc ) of solvent H 3 0 0 are required to manufacture each unit of...
-
This reaction is exothermic. Predict the effect (shift right, shift left, or no effect) of increasing and decreasing the reaction temperature. How does the value of the equilibrium constant depend on...
-
Consider this reaction at equilibrium: Predict whether the reaction will shift left, shift right, or remain unchanged after each disturbance. a. C is added to the reaction mixture. b. H 2 O is...
-
The graphs show that the three components of love, namely, passion, intimacy, and commitment, progress differently over time. Passion peaks early in a relationship and then declines. By contrast,...
-
8.5 moles of an ideal gas (monatomic) expand adiabatically, and performing 8300 J of work in the process. (a) What is the change in internal energy of the gas?. (b) What is the change in temperature...
-
When a 295 g piece of iron at 190C is placed in a 95 g aluminum calorimeter cup containing 250 g of a liquid at 10C, the final temperature is observed to be 38C. Determine the specific heat of the...
-
A glass window is 2.0 m wide and 1.5 m high and 7.5 mm thick. The temperature of the inner surface is 18 C and that of the outer surface is 11 C. (a) Find the rate of heat flow through the window....
-
On a clear day, a person lying on a beach. If the sun rays make an angle of 45 from the vertical, what is the rate of energy absorption from the Sun by the person? Assume the surface area of the...
-
1. Within 5.0 s of lift-off, a space shuttle reaches an altitude of 525 m, uniformly accelerating during the entire time. (a) What is its acceleration? (b) At what speed is the shuttle travelling...
-
The December 31, Year 8, trial balance of SwissCo Ltd., a Swiss company, follows (in euros, ). Additional Information: 1. SwissCo uses the periodic inventory system along with the FIFO costing method...
-
What is EBIT/eps analysis? What information does it provide managers?
-
A system is being designed to carry 500 gal/min of ethylene glycol at 77F at a maximum velocity of 10.0 ft/s. Specify the smallest standard Schedule 40 steel pipe to meet this condition. Then, for...
-
The range of Reynolds numbers between 2000 and 4000 is described as the critical region because it is not possible to predict whether the flow is laminar or turbulent. One should avoid operation of...
-
The water line described in Problem 8.22 was a cold water distribution line. At another point in the system, the same-size tube delivers water at 180F. Compute the range of volume flow rates for...
-
Fosters Manufacturing Co . warrants its products for one year. The estimated product warranty is 2 % of sales. Assume that sales were $ 1 , 8 0 0 , 0 0 0 for January. On February 7 , a customer...
-
The City of Eeyore budget for the fiscal year ended June 3 0 , 2 0 2 0 , included an appropriation for the police department in the amount of $ 8 , 7 1 6 , 0 0 0 . During the month of July 2 0 2 0 ,...
-
Sandia Corporation manufactures metal toolboxes. It adds all materials at the beginning of the manufacturing process. The company has provided the following information: Units Costs Beginning work in...

Study smarter with the SolutionInn App


