Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict
Question:
Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change.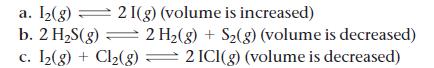
Transcribed Image Text:
a. 1₂(g) 21(g) (volume is increased) b. 2 H₂S(g) = 2 H₂(g) + S₂(g) (volume is decreased) c. I₂(g) + Cl₂(g) 2 ICI(g) (volume is decreased)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
a Shift ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume...
-
Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change. CO(g) + H(g)...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Q1. A 10-kg uniform rod ABC is connected to two collars of negligible mass which slide on horizontal, frictionless rods located in the same vertical plane. If a force P of magnitude 50 N is applied...
-
Drumwright Company needs to increase its profits and so has embarked on a program to increase its overall productivity. After one year of operation, Kent Olson, manager of the Columbus plant,...
-
Write a chemical equation showing the preparation of octadecyl p-toluenesulfonate.
-
How does an Enterprise resolve gaps between a Problem Space and its Solution Space(s)?
-
Ecommerce.com, a large Internet retailer, is studying the lead time (elapsed time between when an order is placed and when it is filled) for a sample of recent orders. The lead times are reported in...
-
Chattanooga Co. has collected the following per unit data: Direct labor $4 Variable selling and $5 admin. Direct materials 6 Fixed overhead $3 Variable overhead 3 Fixed selling and admin. $8 with a...
-
This reaction is exothermic. Predict the effect (shift right, shift left, or no effect) of increasing and decreasing the reaction temperature. How does the value of the equilibrium constant depend on...
-
Consider this reaction at equilibrium: Predict whether the reaction will shift left, shift right, or remain unchanged after each disturbance. a. C is added to the reaction mixture. b. H 2 O is...
-
Determine the differential equations governing the motion of the system by using the equivalent systems method. Use the generalized coordinates shown in Figures P2.59. A -- mim 1 m 12 B 2m Slender...
-
Two resistors are connected in parallel with an 99 volt battery. The resistors have values of R 1 = 7 ohms and R 2 = 55 ohms. What is the current (in amps) in this circuit? Round your answer to two...
-
Blood flows through the aorta at an average speed of v = 19cm/s. The aorta is roughly cylindrical with a radius r = 17.6 mm. The volume rate of blood flow through the aorta is r 2 v . Calculate the...
-
A solenoid has N turns of area A distributed uniformly along its length. When the current in this solenoid increases at the rate of 2.0 A/s, an induced emf of 75 mV is observed. Suppose the spacing...
-
The Royal Gorge Bridge in Colorado rises 321 m above the Arkansas River. Suppose you kick a rock horizontally off the bridge. The magnitude of the rock's horizontal displacement is 45.0 m. Find the...
-
How might they explain what they observed? https://www.youtube.com/watch?v=BoeDI-YkzI0 Construct an explanation for the investigation of sound and vibration shown in the video. You can make your own...
-
Newmont Mining is the largest gold producer in North America and second largest in the world, with mining interests in the U.S., Mexico, Peru, Uzbekistan, and Indonesia. In 1998, Newmont produced...
-
The unadjusted trial balance of Secretarial Services is as follows: SECRETARIAL SERVICES Unadjusted Trial Balance as at 31 December 2017 Account Debit Credit Cash at bank Office supplies Prepaid...
-
In a dairy, milk at 100F is reported to have a kinematic viscosity of 1.30 centistokes. Compute the Reynolds number for the flow of the milk at 45 gal/min through a 1-in steel tube with a wall...
-
In a soft-drink bottling plant, the concentrated syrup used to make the drink has a kinematic viscosity of 17.0 centistokes at 80F. Compute the Reynolds number for the flow of 215 L/min of the syrup...
-
A certain jet fuel has a kinematic viscosity of 1.20 centistokes. If the fuel is being delivered to the engine at 200 L/min through a 1-in steel tube with a wall thickness of 0.065 in, compute the...
-
On April 1 , 2 0 2 2 , Guy Comeau and Amelie Lavoi formed a partnership in Ontario. Comeau Lavoi Contribution $ 2 8 3 , 1 0 0 cash $ 2 1 6 , 0 0 0 land $ 1 4 4 , 9 0 0 building Profit sharing $ 1 7 4...
-
Tanner-UNF Corporation acquired as a long-term investment $260 million of 6% bonds, dated July 1, on July 1, 2024. Company management has classified the bonds as an available-for-sale investment. The...
-
How much will net operating income increase ( decrease ) per month if the monthly advertising budget increases by $ 5 , 0 0 0 , the monthly sales volume increases by 1 0 0 units, and the total...

Study smarter with the SolutionInn App


