Gallium has two naturally occurring isotopes with the following masses and natural abundances: Sketch the mass spectrum
Question:
Gallium has two naturally occurring isotopes with the following masses and natural abundances: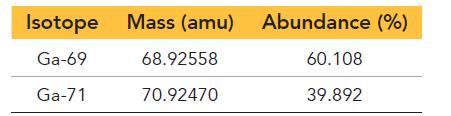
Sketch the mass spectrum of gallium.
Transcribed Image Text:
Isotope Ga-69 Ga-71 Mass (amu) Abundance (%) 60.108 39.892 68.92558 70.92470
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Fig Mass spec...View the full answer

Answered By

Daniel Kimutai
I am a competent academic expert who delivers excellent writing content from various subjects that pertain to academics. It includes Electronics engineering, History, Economics, Government, Management, IT, Religion, English, Psychology, Sociology, among others. By using Grammarly and Turnitin tools, I make sure that the writing content is original and delivered in time. For seven years, I have worked as a freelance writer, and many scholars have achieved their career dreams through my assistance.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Patients seeking care at the County General emergency room wait, on average, 6 minutes before seeing the triage nurse who spends, on average, 4 minutes assessing the severity of their problem. The...
-
Magnesium has three naturally occurring isotopes with the following masses and natural abundances: Sketch the mass spectrum of magnesium. Isotope Mg-24 Mg-25 Mg-26 Mass (amu) 23.9850 24.9858 25.9826...
-
An element has two naturally occurring isotopes with the following masses and abundances: Isotopic Mass (amu) Fractional Abundance 49.9472. 2.500 103 50.9440. 0.9975 What is the atomic mass of this...
-
Draw structures for the following molecules (a) Acrylonitrile, C3H3N, which contains a carbon-carbon double bond and a carbon-nitrogen triple bond (b) Ethyl methyl ether, C3H8O, which contains an...
-
Rainbow Tours gives walking tours of Springfield. Rainbow charges $40 per person for the tour and incurs $16 in variable costs for labor, drinks, and maps. The monthly fixed costs for Rainbow Tours...
-
Consider a Carnot-cycle heat pump with R-22 as the working fluid. Heat is rejected from the R-22 at 100 F, during which process the R-22 changes from saturated vapor to saturated liquid. The heat is...
-
Estimate the overall odds ratio of the set of tables in Problem3.6 and test whether the odds ratios are the same across the tables. Problem3.6 is: 3.6 Use the DOS data to test whether there is gender...
-
The following transactions apply to Comfort Mattress Sales for 2010. 1. The business was started when the company received $30,000 from the issue of common stock. 2. Purchased mattress inventory of...
-
Differentiate between Market and Book Value of an asset. Define how the Book Value of an asset is calculated for tax purposes. Explain how the market or sales value of an asset might be estimated.
-
Toronto Cricket Property Inc. (TPI) has been incorporated with the purpose of build and operate a world-class cricket stadium outside Toronto. The land on which the stadium will be build is currently...
-
Which pair of elements do you expect to be most similar? Why? a. Nitrogen and oxygen b. Titanium and gallium c. Lithium and sodium d. Germanium and arsenic e. Argon and bromine
-
Which pair of elements do you expect to be most similar? Why? a. N and Ni b. Mo and Sn c. Na and Mg d. Cl and F e. Si and P
-
When a raindrop falls, it increases in size and so its mass at time t is a function of t, namely m(t). The rate of growth of the mass is km(t) for some positive constant k. When we apply New ton's...
-
Given the following diagram: a) Prove that AABC is similar to AADE ABC is ZB of the large triangle. Middle letter indicates the angle within the triangle indicated using the three letters. 6 cm b)...
-
Exercise 4. Company XYZ is evaluating the feasibility of manufacturing a new product. The plant it owns has the capacity to produce 1,000,000 units of the product. Fixed costs for this initiative...
-
Skippy wants to have $17,000.00 in 10 years. His bank is offering an account that earns 1% compounded monthly. How much does he need to deposit to reach his goal? Round your final answer up to the...
-
Rachel wants to have $3,600.00 in 36 months. Her bank is offering her a Certificate of Deposit, a special savings account, that earns 2.3% compounded weekly. How much does she need to deposit now to...
-
Fresh Ltd has two retail businesses that represent separate cash generating units, Fresh Juice Bar and Fresh Salads. At 30 June 2019, the carrying amounts of the assets of the units, valued pursuant...
-
Rainger Company manufactures DVD players and sells them for $125 each. According to the companys records, the variable costs, including direct labor and direct materials, are $45. Factory...
-
What can scientists learn by comparing the fossilized skeletons of extinct primates with the bones of modern species?
-
There are two different compounds with molecular formula C 2 H 6 O. One of these isomers has a much higher boiling point than the other. Explain why.
-
Determine the relationship between the two structures below. Are they resonance structures or are they constitutional isomers?
-
In each reaction, identify the Lewis acid and the Lewis base: (a) (b) (c) F L-
-
How does socialization contribute to the formation and perpetuation of cultural norms and values within a society ?
-
Vaughn Company reported the following amounts in the stockholders ? ?equity section of its December 3 1 , 2 0 2 4 , ?balance sheet. Preferred stock, 1 0 % , ?$ 1 0 0 ?par ( 1 0 , 0 0 0 ?shares...
-
On August 31, 2021, Brooke Tuck borrowed $6,000 from Commonwealth State Bank. Tuck signed a note payable, promising to pay the bank principal plus interest on August 31, 2022. The interest rate on...

Study smarter with the SolutionInn App


